 |
 |
 |
| |
The ADVANCE trial: Phase 3, randomized comparison of TAF/FTC/DTG, TDF/FTC/DTG or TDF/FTC/EFV for first-line treatment of HIV-1 infection
|
| |
| |
Reported by Jules Levin
IAS 2019 Mexico City July 22-25
IAS: Progressive rises in weight and clinical obesity for TAF/FTC+DTG and TDF/FTC+DTG versus TDF/FTC/EFV: ADVANCE and NAMSAL trials - (07/25/19)

We performed multivariate analyses to adjust for various factors, including
1.Sociodemographic factors: age, gender, nationality, relationship status, education level, employment status
2.BL factors: weight/BMI, treatment arm, CD4 count and VL
3.Disease history and adverse events: history of HPT, DM or dyslipidaemia
4.Concomitant medications: contraceptives, amlodipine, psychotropic medications and prednisone
Reported by Jules Levin
IAS 2019 Mexico City July 22-25
Dolutegravir plus two different formulations of tenofovir, and emtricitabine, for the treatment of HIV-1 infection: ADVANCE trial
W.F. Venter1, M. Moorhouse1, S. Sokhela1, L. Fairlee1, N. Mashabane1, M. Masenya1, A. Qavi2, P. Clayden3, E. Abrams4, B. Simmons2, A. Hill5
1University of Witwatersrand, Health Sciences, Johannesburg, South Africa, 2Imperial College, Faculty of Medicine, London, United Kingdom, 3HIV i-Base, London, United Kingdom, 4Columbia University, ICAP, Maidman School of Public Health, New York, United States, 5University of Liverpool, Translational Medicine, Liverpool, United Kingdom
Background: In low- and middle-income countries, most treatment-naïve people living with HIV (PLWH) take tenofovir disoproxil fumarate (TDF) with FTC (or 3TC) and efavirenz (EFV). Dolutegravir (DTG) and tenofovir alafenamide fumarate (TAF) are recommended in international guidelines, but clinical experience with these ARVs in sub-Saharan Africa is limited. In South Africa, over 10% of patients have transmitted NNRTI drug resistance.
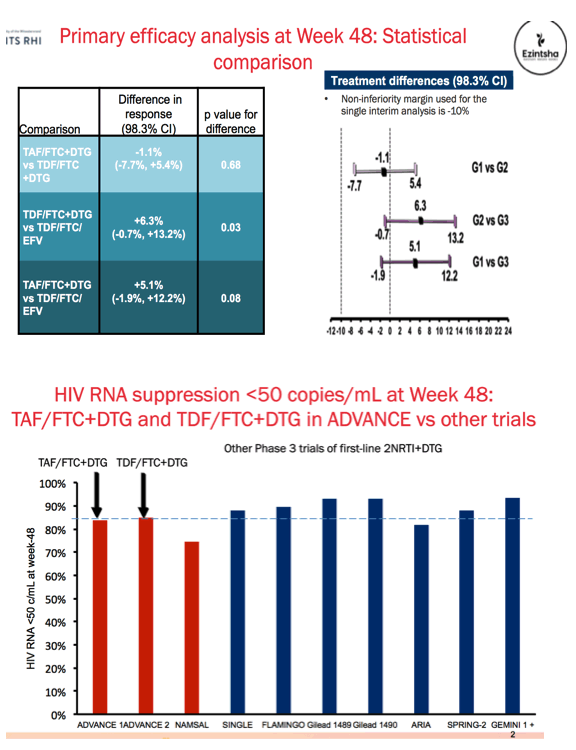
Methods: We conducted a 96-week, open-label randomised trial in South Africa, comparing TAF/FTC/DTG, TDF/FTC/DTG and TDF/FTC/EFV. Inclusion criteria included age ≥12 years, no prior ART >30 days, creatinine clearance >60 mL/min (>80 mL/min if <19 years), and HIV-1 RNA >500 copies/mL. Pregnancy and tuberculosis (TB) were exclusion criteria. There was no screening for baseline drug resistance, consistent with South African treatment guidelines. The primary treatment failure endpoint was 48-week HIV-1 RNA >50 copies/mL, discontinuation or missing data (Intent-to-treat population, non-inferiority margin -10%, significance level p=0.017, adjusted for multiple comparisons). We report 48-week efficacy and safety data.
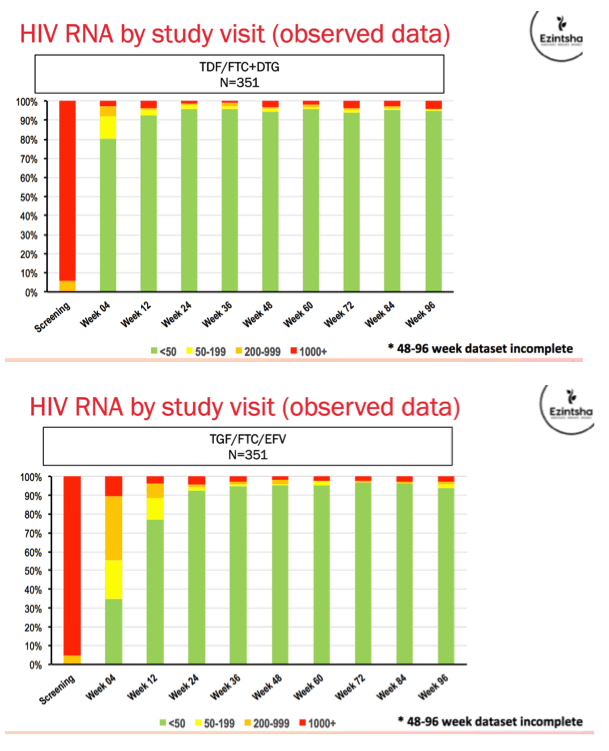
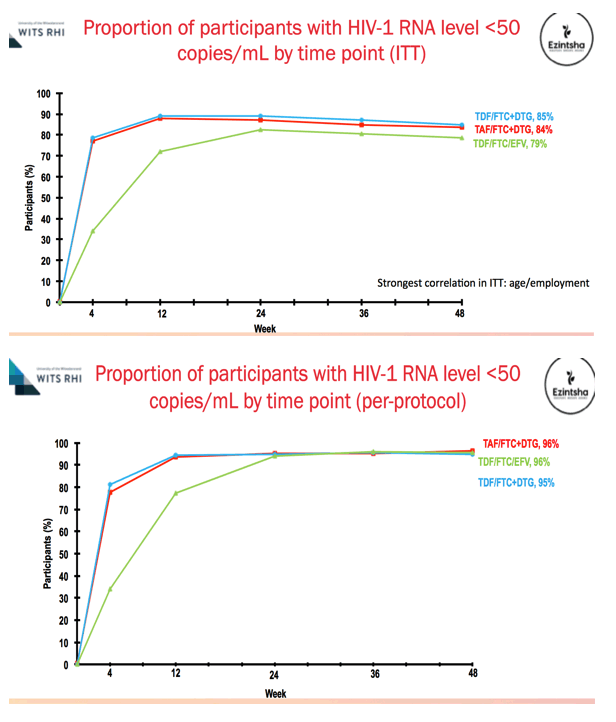
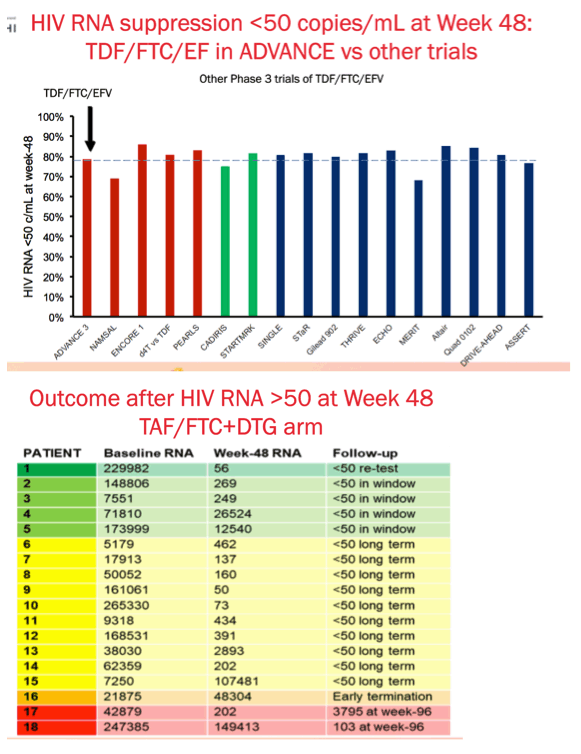
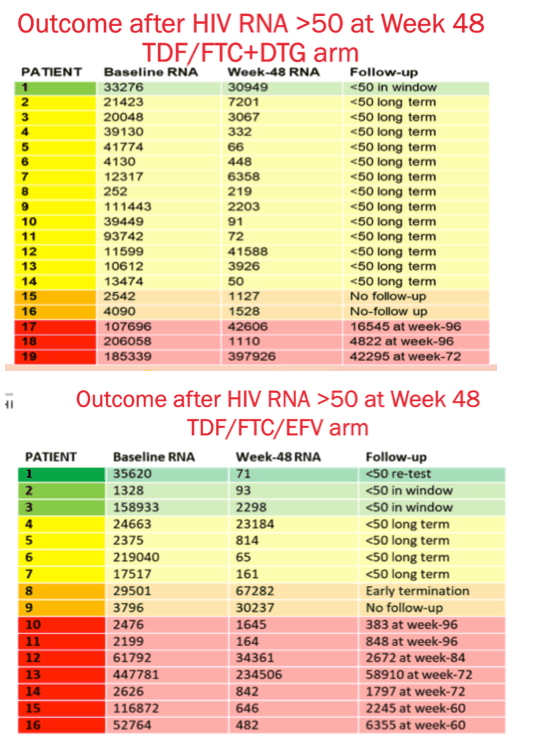
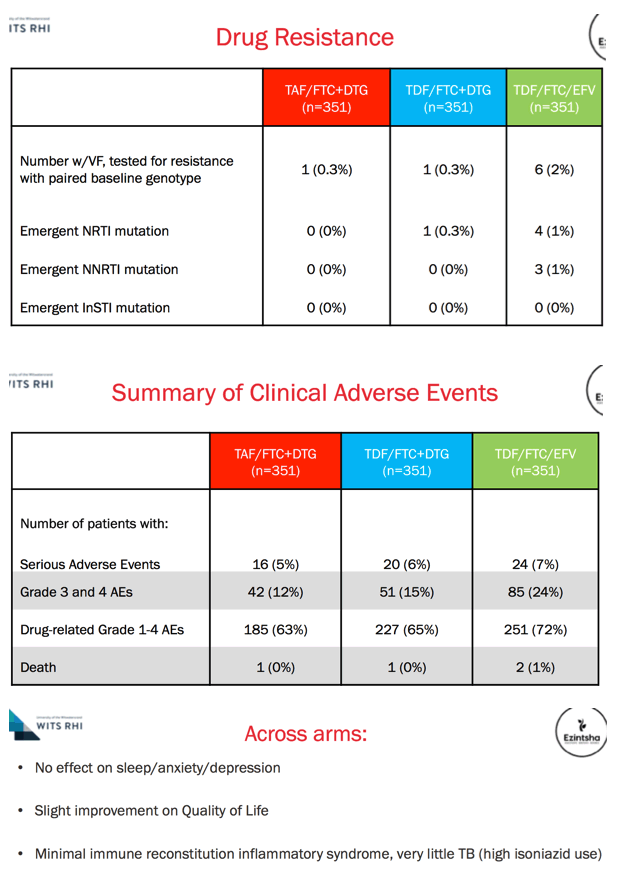
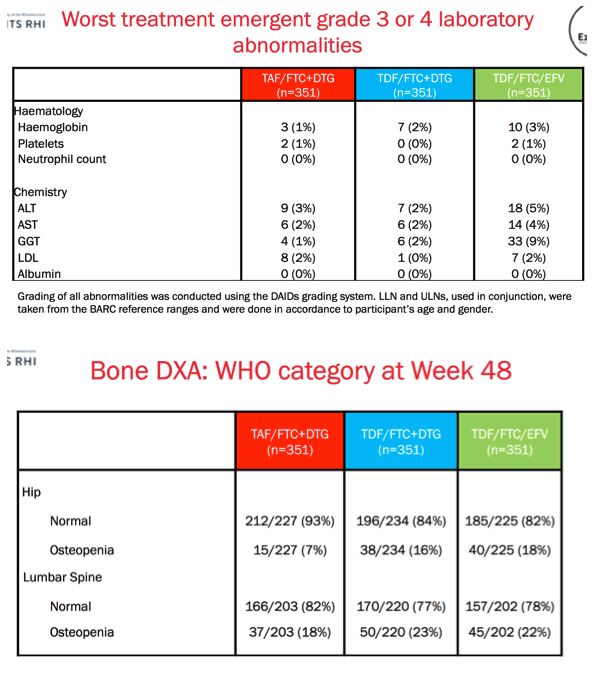
Results: We randomised 1053 PLWH between February 2017 and May 2018: 99% black, 59% female, mean age 32 years, with mean CD4 336 cells/uL. At week 48, the percentage of participants with HIV RNA < 50 copies/mL was 83.8% for TAF/FTC/DTG, 84.9% for TDF/FTC/DTG and 78.6% for TDF/FTC/EFV. In the on-treatment analysis, 96% of participants on TAF/FTC/DTG, 94% on TDF/FTC/DTG and 95% on TDF/FTC/EFV had HIV RNA < 50 copies/mL at Week 48. Both DTG arms demonstrated non-inferior efficacy versus the EFV arm. Over 70% of participants with HIV RNA >50 copies/mL re-supressed after adherence counselling and re-testing. Overall, 136/185 (74%) of treatment failures were from discontinuation. Clinical adverse events and laboratory abnormalities were similar between treatment arms.
Conclusions: In the ADVANCE study, TAF/FTC/DTG and TDF/FTC/DTG demonstrated non-inferior efficacy versus TDF/FTC/EFV, with low rates of virologic failure in all three arms despite country-level background NRTI/NNRTI resistance. There were more discontinuations for adverse events in the TDF/FTC/EFV arm.


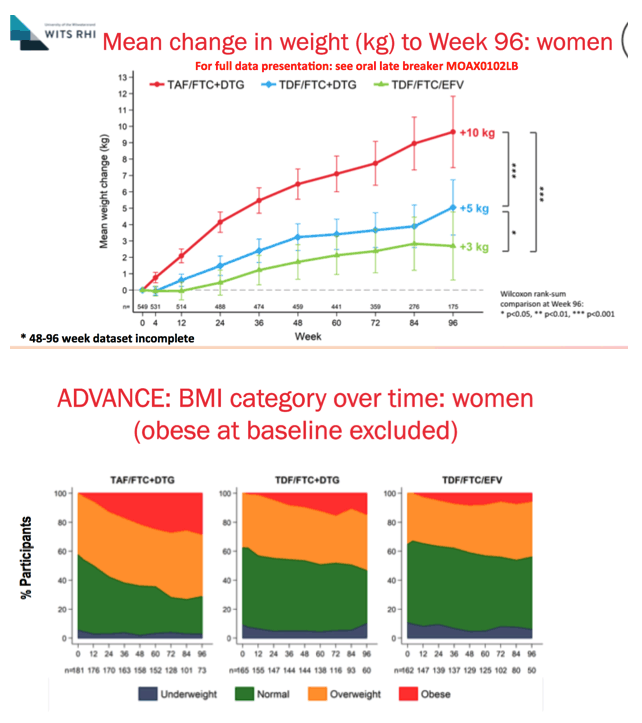
Here we show changes in BMI category over 96 weeks for women in ADVANCE, with those who were obese at BL excluded from the analysis. More women receiving TAF/FTC+DTG became overweight or obese than the other 2 study arms: by week 96, almost 70% of women in the TAF/FTC+DTG were overweight or obese. More than 20% of women in the TAF/FTC+DTG arm became obese by week 96, compared to less than 20% in the TDF/FTC+DTG arm and less than 10% in the EFV-containing arm. There were more underweight women in the two TDF-containing arms, and this was relatively stable across the study.
At the time of the analysis, not all participants had reached week 96 of the study; accordingly the number of participants at each time point is represented.
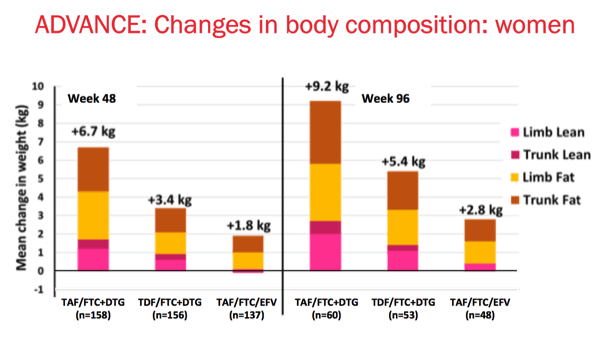
Looking at the DEXA data in women: Increases in trunk and limb fat were higher in both DTG arms and increased further from week 48 to week 96 in women. Increases in both limb and trunk fat which were considerably higher in women on the TAF/FTC+DTG arm and which continued to increase substantially from week 48 to week 96. Increases in lean mass, both limb and trunk, were also higher in the DTG arms than with EFV.
In the EFV arm, we can see different changes, with smaller increases in limb and trunk fat, and minimal increase in lean mass by week 96.
At the time of the analysis, not all participants had reached week 96 of the study; accordingly the number of participants at each time point is represented.


|
| |
|
 |
 |
|
|