 |
 |
 |
| |
ON-DEMAND HIV POSTEXPOSURE PROPHYLAXIS BY TAF/EVG VAGINAL INSERTS IN MACAQUES
|
| |
| |
CROI 2020 - see blow 2019 PrEP talk & 2020 PEP oral.
Reported by Jules Levin
Charles Dobard1, M. Melissa Peet2, Kenji Nishiura1, Angela Holder1, Onkar N. Singh2, Timothy J. McCormick2, James Mitchell1, George Khalil1, Gerardo Garcia-Lerma1
1CDC, Atlanta, GA, USA,2CONRAD, Arlington, VA, USA,3University of the Sciences, Philadelphia, PA, USA
Abstract
Topical products for HIV prevention that can be administered on-demand, either pre- or post-viral exposure, remain an advantageous regimen for end-users that have infrequent or clustered intercourse or are unable to take daily pills. CONRAD has developed a topical insert co-formulated with tenofovir alafenamide fumarate (TAF) and elvitegravir (EVG) to provide end-users a flexible on-demand dosing regimen that may align with their needs and lifestyle. Recently, we showed in a macaque model that mimics vaginal HIV transmission in women that a vaginal insert containing a fixed-dose combination of TAF and EVG was highly effective in preventing SHIV infection when administered 4 hours before virus exposure. Here, we investigated the efficacy of the same TAF/EVG insert when administered 4 hours after SHIV exposure.
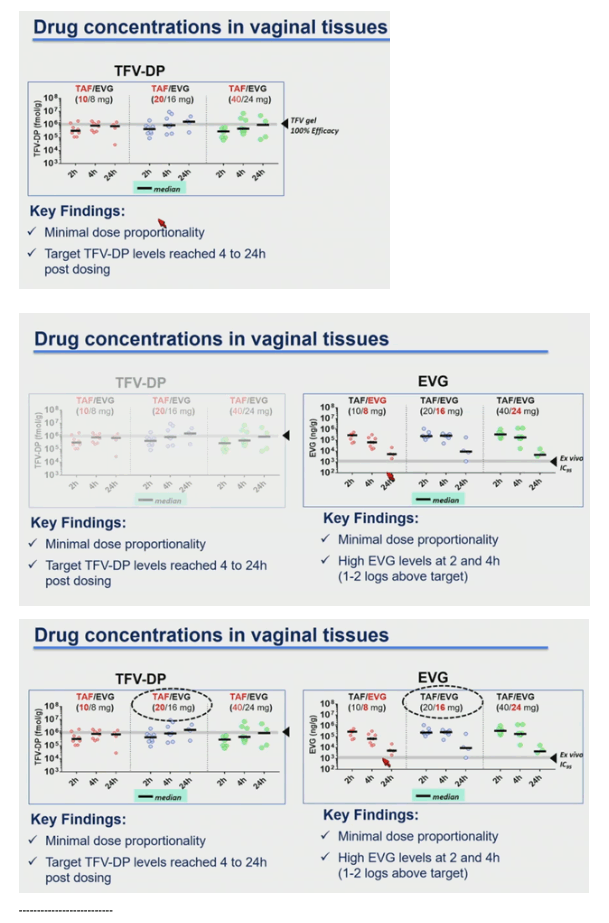
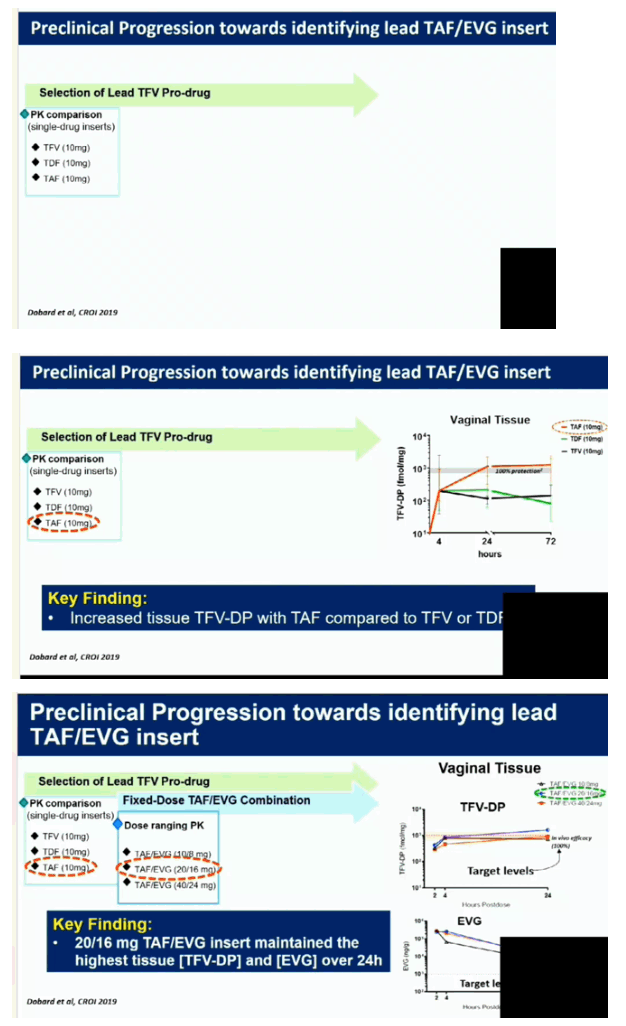
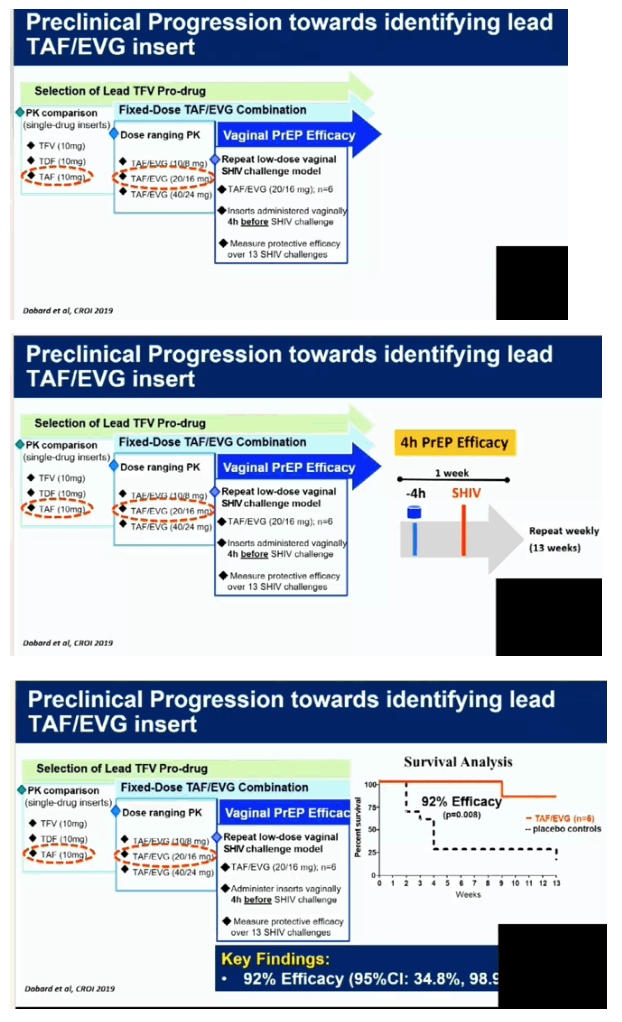
Normal cycling pigtail macaques (n=11) were exposed vaginally to SHIV162P3 once-weekly for 13 weeks. Six macaques received TAF/EVG inserts (20/16 mg) and 5 received a placebo. Inserts were placed in the posterior vagina near the cervix 4 hours after each SHIV exposure. Infection was monitored weekly by serology and RT-PCR of SHIV RNA in plasma. The concentrations of TAF, TFV, and EVG in plasma and TFV-DP in PBMCs were measured at 4 hours in a second group of 6 macaques that received the same TAF/EVG inserts once-weekly for 13 consecutive weeks.
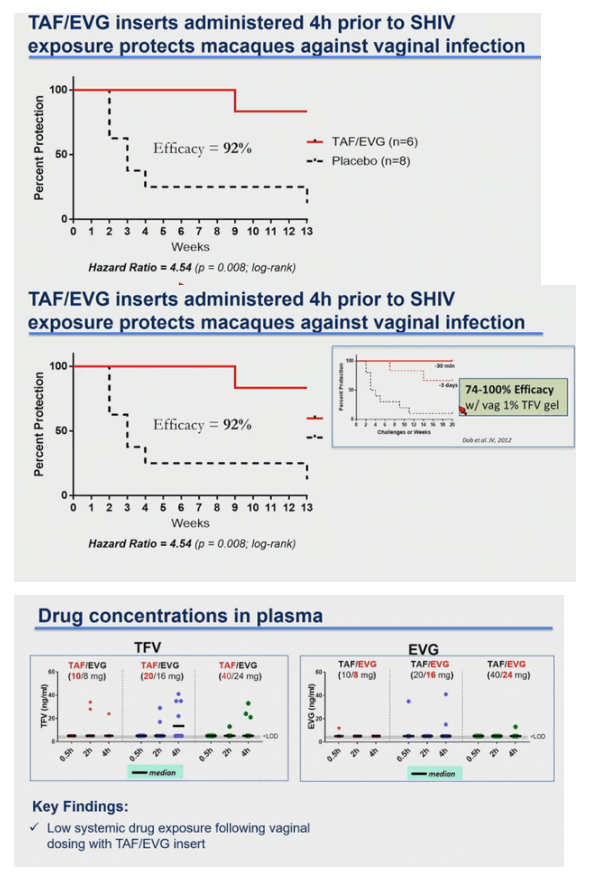
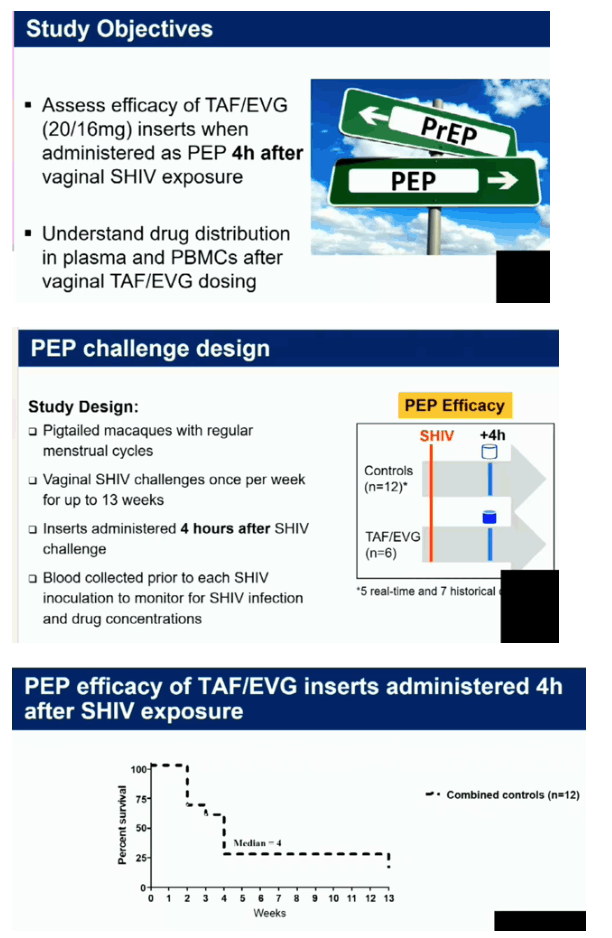
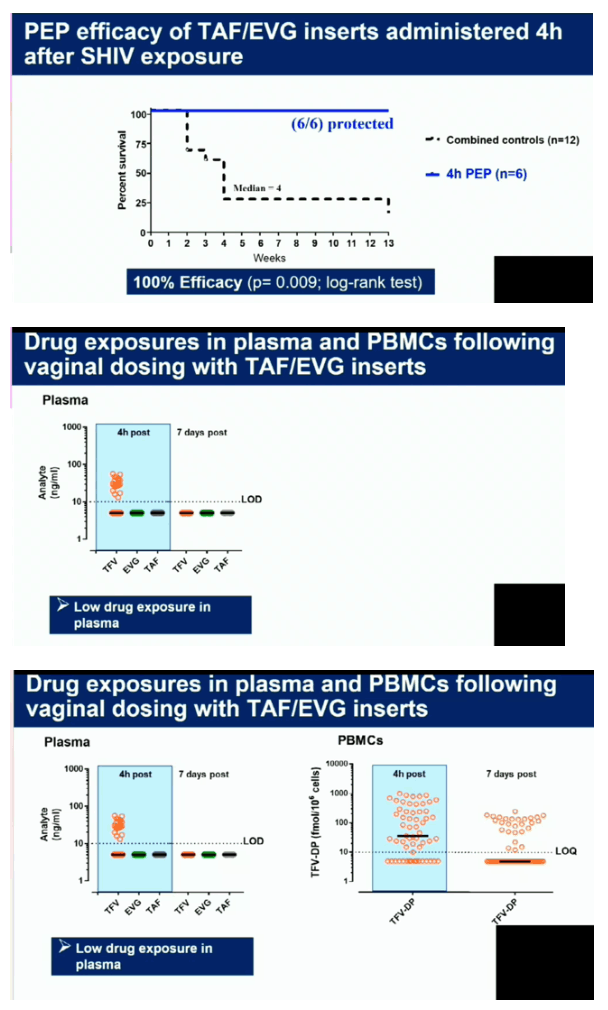
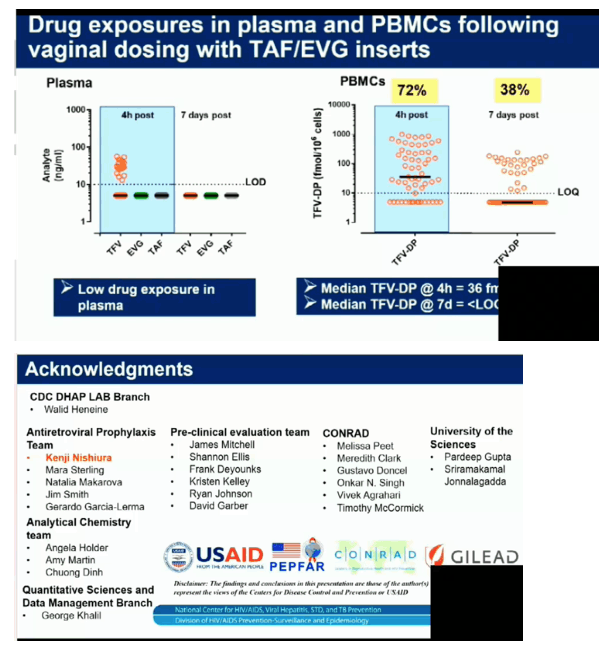
Four of the 5 macaques that received placebo inserts became infected with SHIV after a median of 4 challenges (range 2-13). In contrast, all 6 macaques that received TAF/EVG inserts 4h after SHIV exposure remained protected after 13 challenges and a 20-week follow-up period (p=0.009; log-rank test). The calculated PEP efficacy of TAF/EVG inserts was 100%. Of the 78 plasma specimens collected 4h post insert dosing, EVG was only detected in 1 sample (15 ng/ml); none had detectable TFV or TAF. Conversely, TFV-DP was detected in 42/59 PBMC samples; median level in samples with detectable TFV-DP was 147.5 [range=15-993] fmol/106 cells.
Vaginal administration of a single TAF/EVG insert several hours after virus exposure fully protected macaques against SHIV infection, thus increasing flexibility and expanding our established window of protection to 4 hours before or after sex. The observed high levels of TFV-DP in PBMCs by topical delivery of TAF is unique and may have contributed to protection. Our data support the clinical development of TAF/EVG inserts for on-demand PrEP/PEP for HIV prevention.
http://www.croiwebcasts.org/console/player/44700?mediaType=slideVideo&
2019 oral: http://www.croiwebcasts.org/console/player/41207?mediaType=slideVideo&
Vaginal administration of inserts containing TAF and EVG was highly effective in preventing SHIV infection in a macaque model that mimics vaginal transmission of HIV in women. The data support the clinical development and first-in-human testing of TAF/EVG inserts for on-demand topical prophylaxis against vaginally acquired HIV infection.



|
| |
|
 |
 |
|
|