 |
 |
 |
| |
B/F/TAF Controls HIV in 10 of 10 on Hemodialysis for Kidney Disease
|
| |
| |
IDWeek 2020, October 22-25, 2020
Mark Mascolini
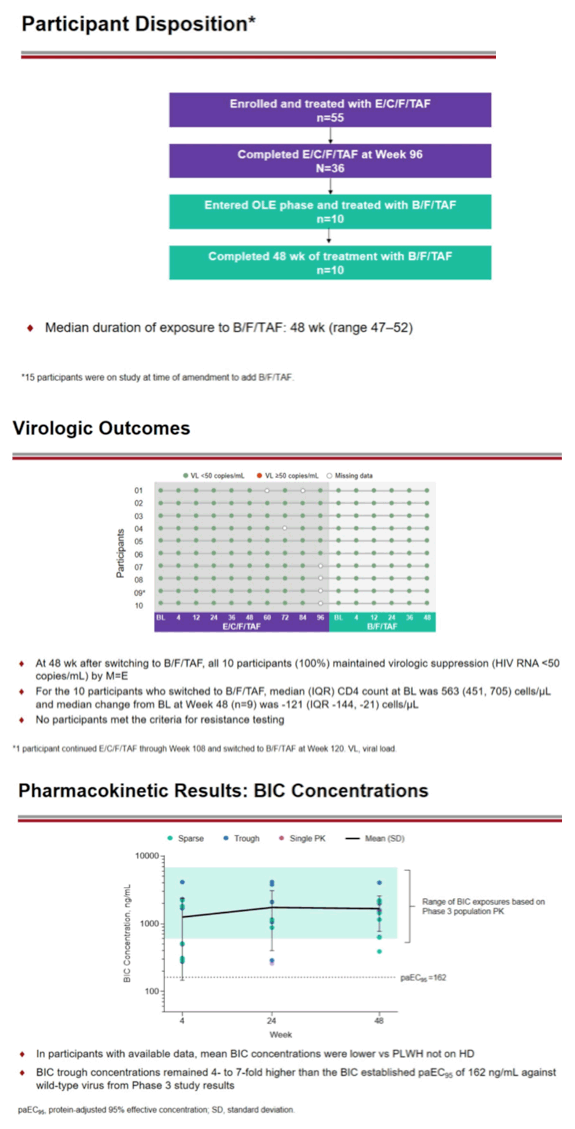
Through 48 weeks 10 of 10 people receiving chronic hemodialysis for end-stage renal disease (ESRD) maintained a viral load below 50 copies in an open-label extension study of B/F/TAF, the single-tablet once-daily regimen combining the integrase inhibitor bictegravir with emtricitabine (FTC) and tenofovir alafenamide (TAF) [1]. No one stopped taking B/F/TAF during this analysis.
The study involved people on hemodialysis with ESRD (serious kidney disease) taking the single-tablet combination elvitegravir/cobicistat/F/TAF (E/C/F/TAF) [2]. After analysis of phase 3 B/F/TAF data and licensing of this single-tablet therapy by the FDA, these people were allowed to replace the elvitegravir combination with B/F/TAF.
Ten of 36 people made the change, all of them living in the United States. Eight were men and 9 were black. All had a viral load below 50 copies, and median CD4 count stood at 563. They had spent a median of 4 years receiving hemodialysis, and median estimated glomerular filtration rate measured 11.6 mL/min. All 10 people had hypertension, 6 had cardiovascular disease, 4 had diabetes, and 3 had hyperlipidemia.
All 10 people completed 48 weeks of B/F/TAF therapy. Six of 10 people with viral load data after taking E/C/F/TAF for 96 weeks had a viral load below 50 copies. When checked at the start of B/F/TAF, all 10 study participants had a viral load below 50 copies, and all 10 maintained that level at weeks 4, 12, 24, 36, and 48 of follow-up. Median CD4 count for the group fell from 563 to 442.
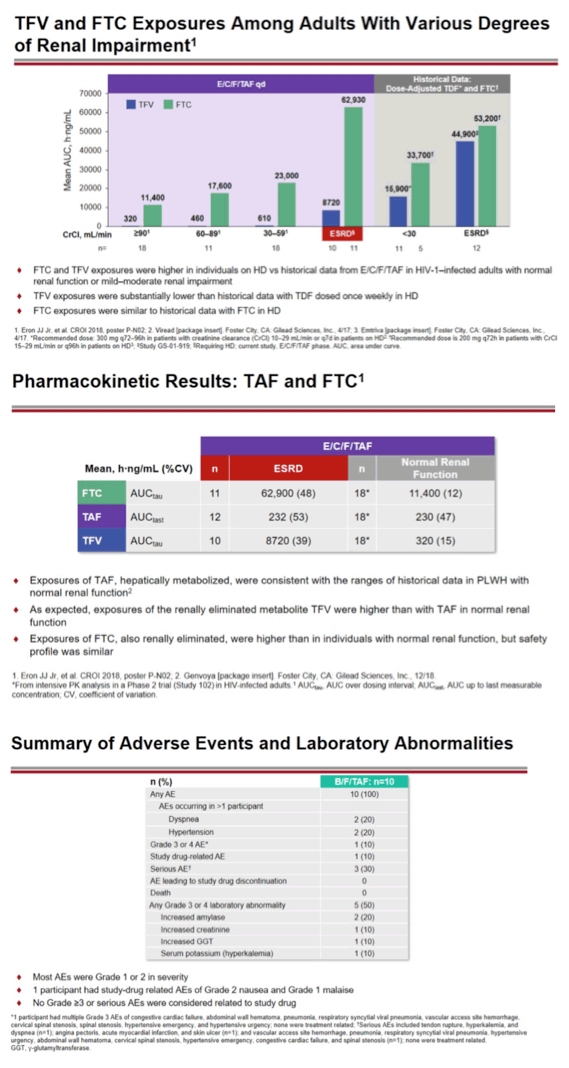
Average bictegravir concentrations during these 48 weeks were lower than in HIV-positive people not receiving hemodialysis. But bictegravir trough concentrations remained 4- to 7-fold higher than the bictegravir protein-adjusted 95% effective concentration of 162 ng/mL against nonresistant virus. FTC and tenofovir* concentrations were higher with B/F/TAF in these hemodialysis patients than in people taking E/C/F/TAF with normal kidney function or mild to moderate kidney impairment. FTC levels were similar to those in hemodialysis patients who had taken FTC in the past. Tenofovir* levels with B/F/TAF were substantially lower than in hemodialysis recipients who had taken tenofovir disoproxil fumarate (TDF) once weekly.
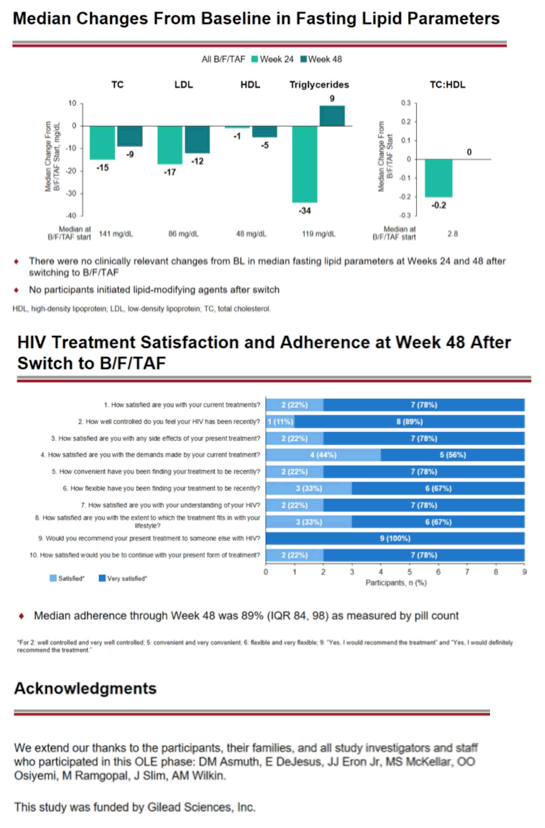
One of 10 people had grade 3 or 4 adverse events while taking B/F/TAF, but these events were not judged related to study drugs. One person had study drug-related adverse events (grade 2 nausea and grade 1 malaise), and 3 had serious adverse events not related to study drugs. No one had an adverse event leading them to stop B/F/TAF. No one had clinically relevant changes in fasting lipids through 48 weeks of follow-up. No one started a lipid-modifying drug.
Through week 48 median pill-taking adherence measured by pill count stood at 89%. Among 9 people who answered a survey about taking B/F/TAF, 2 reported being satisfied with treatment and 7 reported being very satisfied. Two said they were satisfied with continuing B/F/TAF therapy and 7 said they were very satisfied.
The authors conclude that simplifying antiretroviral therapy to B/F/TAF "offers an effective, safe, and convenient once-daily option for people living with HIV on hemodialysis, who often have complicated HIV dosing schedules, multiple comorbidities, and a high pill burden."
*Tenofovir is the active form of TAF and TDF that is measured in drug-concentration studies.
References
1. Eron JJ, Wilkin A, Ramgopal M, et al. A daily single tablet regimen (STR) of bictegravir/emtricitabine/ tenofovir alafenamide (B/F/TAF) in virologically-suppressed adults living with HIV and end stage renal disease on chronic hemodialysis. IDWeek 2020, October 22-25, 2020. Abstract 1002.
2. ClinicalTrials.gov. Safety, tolerability, pharmacokinetics, and efficacy of E/C/F/TAF fixed dose combination (FDC) in HIV-1 infected adults on chronic hemodialysis. ClinicalTrials.gov identifier NCT02600819. https://clinicaltrials.gov/ct2/show/NCT02600819


|
| |
|
 |
 |
|
|