 |
 |
 |
| |
WEEKLY ORAL TENOFOVIR ALAFENAMIDE PROTECTS MACAQUES FROM VAGINAL SHIV INFECTION
|
| |
| |
CROI 2021 March 6 Reported by Jules Levin
Ivana Massud1, Kenji Nishiura1, Susan Ruone1, Angela Holder1, Chuong Dinh1, Shanon Ellis1, Kristen Kelley1, George Khalil1, Walid Heneine1, Gerardo Garcia- Lerma1, Charles Dobard1
1Centers for Disease Control and Prevention, Atlanta, GA, USA
Background: Poor adherence to daily pre-exposure prophylaxis (PrEP) reduces efficacy and public health benefit. Simpler oral regimens providing long-lasting protection for one or more weeks may be more desirable among people who have difficulties adhering to a daily pill. Here, we report the pharmacokinetic (PK) assessment of a weekly oral tenofovir alafenamide (TAF) regimen in pigtailed macaques and its efficacy against vaginal SHIV infection.
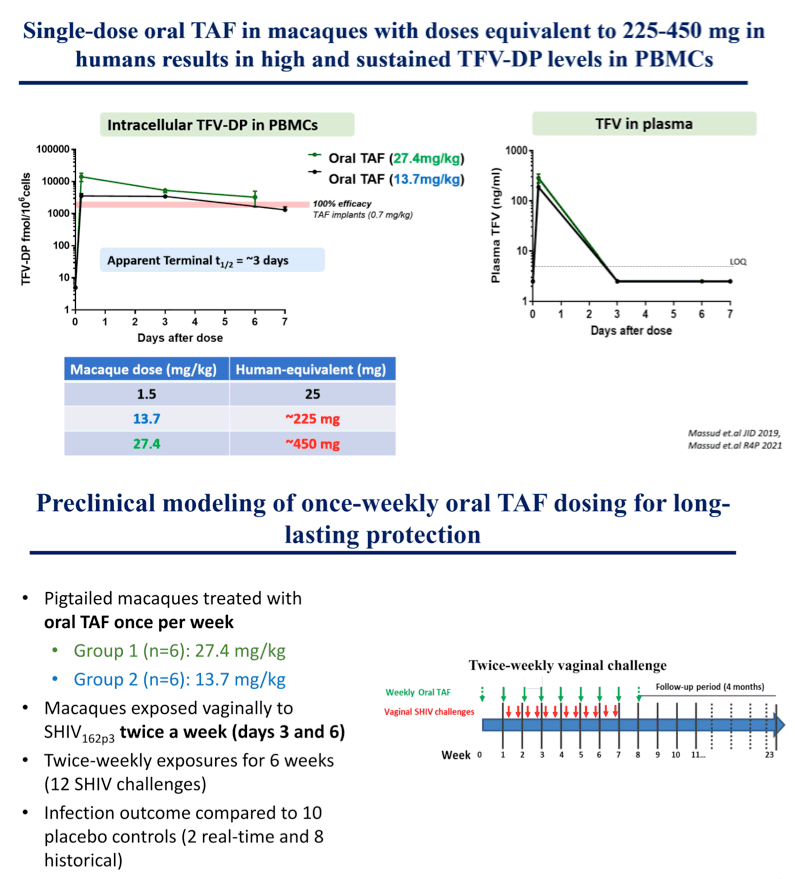
Methods: We previously defined the human equivalent dose of oral TAF and TDF in macaques to be 1.5 and 22 mg/kg, respectively. Here we assessed in macaques the PK profile of a higher dose of TAF (27.4 mg/kg) in plasma and PBMCs following a single oral dose. Efficacy was determined in macaques that received a weekly 27.4 mg/kg dose of oral TAF and were vaginally exposed to SHIV162p3 at 3- and 6-days post dosing for up to 6 weeks (12 challenges). Infection outcome was compared to 10 untreated macaques. Tenofovir (TFV) and TFV diphosphate (TFV-DP) were measured by LC-MS/MS. SHIV RNA was monitored weekly in plasma by RT-PCR.
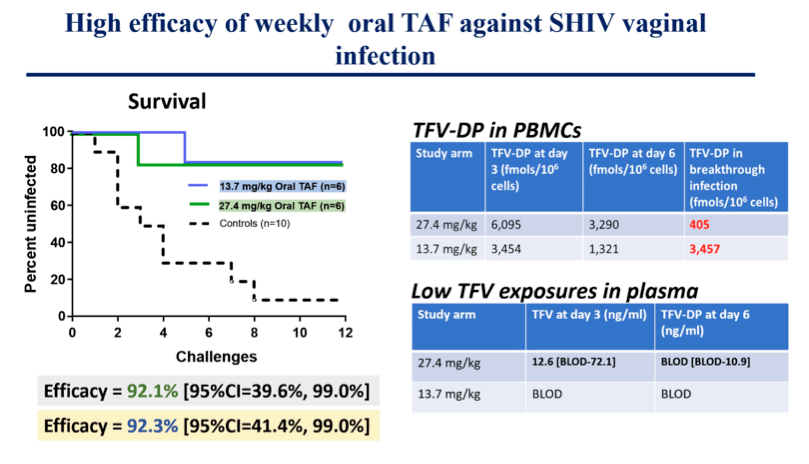
Results: Median TFV levels in plasma at 5h were 284 ng/ml (range 227-341). TFV-DP levels in PBMCs (fmols/106 cells) were 14,090, 6,740, and 4,390 at 5h, 3 days, and 6 days, respectively. Nine of 10 untreated controls were infected after a median of 3 SHIV exposures (range 1-12). In contrast, 5/6 animals receiving a single dose of oral TAF were protected against vaginal SHIV infection (Efficacy = 92.1% [95%CI=39.6%, 99.0%]. Median TFV-DP levels in the protected animals at time of challenge (day 3 and 6) were 6,095 and 3,485 fmols/106 cells, respectively. In contrast, the PrEP breakthrough animal showed much lower TFV-DP in PBMCs at challenges prior to infection (median = 405 [274-677] fmols/106 cells). Using in vivo Cmax TFV-DP levels and calculated TFV-DP half-life in PBMCs of 5.3 days, we estimate the length of prophylactic window will extend well beyond 1 week.
Conclusion: We identified a dose of oral TAF that resulted in high and sustained TFV-DP levels in PBMCs and protected against vaginal SHIV infection for at least 1 week following a single oral administration. The data open the possibility for long-lasting PrEP protection with infrequent oral dosing.


|
| |
|
 |
 |
|
|