 |
 |
 |
| |
Summary from virtual CROI 2021 for HIV and liver disease
HIV and the liver: hot topics
|
| |
| |
Jurgen K. Rockstroh M.D., Professor of Medicine
University of Bonn, Germany
Correspondence:
Prof. Dr. Jurgen K. Rockstroh
Department of Medicine I
University Hospital Bonn
Venusberg-Campus 1
53127 Bonn
Germany
Introduction
2021 already becomes the second year in a row where CROI had to take place as a virtual meeting. The ravaging spread of COVID-19 globally has severely impacted the content of CROI with around 50% of the abstract submissions dealing with research findings around COVID-19. Abstract submissions from other areas of interest such as HIV and liver disease were considerably lower but still many high-quality submissions were made, resulting in an oral coinfection session, which was split between hepatitis and tuberculosis submissions and quite a number of very interesting science spotlight presentations focusing on various aspects of liver disease in HIV. Abstracts are available within the abstract book on the CROI website and have been published in Topics in Antiviral Medicine, Volume 29 Issue 1. Nearly all talks were streamed in real time, and oral presentations are now available as webcasts (see www.croiconference.org). The oral hepatitis session included three presentations this year including results from a very impressive HCV treatment trial with minimal monitoring, an analysis from the NA-ACCORD study investigating the relationship between HBV viremia and risk for hepatocellular cancer in HIV/HBV coinfected patients and finally a lab study looking at circulation of HBV-complex profiles with HBs-Ag vaccine escape mutations (1-3). The former poster sessions from the much-missed face-to-face CROI meetings were transferred into an interactive format named science spotlight, which included the pdf of the respective presentation as well as a prerecorded small summary podcast of the respective research and an interactive platform for questions. These spotlights covered numerous aspects around hepatitis A-D as well as issues around fatty liver disease in HIV (4-18). This report aims at summarizing the main findings around viral hepatitis coinfection but also on new data around fatty liver disease in HIV-infected individuals.
Hepatitis A
One of the science spotlight sessions covered aspects around hepatitis A vaccination in HIV-infected individuals. This is a very important topic in light of the recent hepatitis A outbreaks both worldwide and in the United States. Most national and international HIV management guidelines clearly recommend hepatitis A, B and C screening upon first visit to an HIV-clinic. Patients with a negative HAV serology are recommended to receive hepatitis A vaccination. At CROI 2021 data from a retrospective chart review of HAV vaccination rates in people living with HIV who attended 2 clinics in Houston, Texas between January 1, 2010 and December 31, 2019 was presented (4). Patients were considered eligible for HAV vaccine if they were HAV IgG negative or never had this test performed and never received prior HAV vaccination. 36.9 % of MSM (n=1135) were eligible for hepatitis A vaccination Within this analysis the cumulative incidence of receiving 1 and 2 doses of hepatitis A vaccine at 6, 12 and 24 months from entry to HIV care was determined. The corresponding results are shown in Table 1.
Table 1: Cumulative incidence of receiving at least 1 HAV vaccination
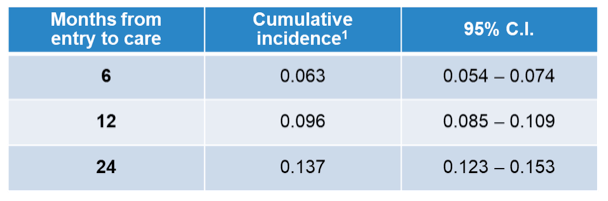
The cumulative incidence of receiving a second HAV vaccination is shown in table 2.
Table 2: Cumulative incidence of receiving 2 HAV vaccination,
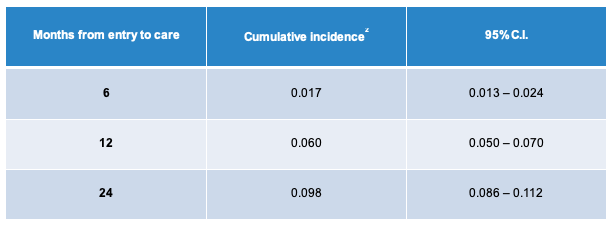
Overall, of 6,515 patients, 3,074 were eligible for HAV vaccination. 6.3% of patients received at least 1 HAV vaccine within 6 months from entry to care, 9.6% received 1 vaccine within 12 months and 13.7% received at least 1 vaccination within 24 months. MSM and patients with chronic HBV and/or C were more likely to receive 1 and 2 vaccines, whereas the Hispanic population was less likely to receive 1 and 2 vaccinations. Clearly, vaccination rates are too low and programs to better close vaccination gaps are urgently needed.
Similar findings were reported from a Spanish study from Sevilla which demonstrated that in 270 HAV susceptible HIV patients (30%) 62% were not HAV vaccinated (5).
After primary hepatitis A vaccination, people living with HIV have a lower serologic response rate and are more likely to experience loss of seroprotective antibodies during follow-up compared to HIV-negative controls. However, it is not clear as to how HAV revaccination should be administered among PLWH who are non-responders or have lost seroprotective antibodies (seroreverters) after primary vaccination. In a further study from Taiwan investigators studied HAV vaccination responses in an open-label randomized clinical
trial which enrolled PLWH who tested negative for anti-HAV antibodies ≥4 weeks after completing primary 2-dose HAV vaccination and those who seroreverted after having had seroresponses to primary vaccination (6). Stratified by CD4 count, all subjects were randomized at a 1:1 ratio to receive either 1 or 2 doses (4 weeks apart) of HAV vaccine with a block size of 4. At week 4, week 8 (only in 2-dose group), week 24, and week 48 after HAV revaccination, the levels of anti-HAV IgG were determined with the use of a semiquantitative chemiluminescence immunoassay (ARCHITECT HAVAb-IgG, Abbott, Germany). A hundred and two participants, 50 in the 2-dose and 52 in the 1-dose group, had completed follow-up at week 24. All participants were male (mean age, 38.6 years), with median CD4 of 461 cells/mm³ and plasma HIV RNA <20 copies/ml in 90.2% before revaccination. The baseline characteristics were well balanced between the two groups. The serologic responses at week 24 was similar: 84.0% for the 2-dose revaccination group and 78.8% for the 1-dose group (difference, 5.2%; 95% CI, -9.9%-20.2%), respectively. Of note, 46.2% of the subjects in the 1-dose arm and 44% of the 2-dose arm had previously developed HAV antibodies after HAV vaccination, respectively. The duration from last HAV vaccination was 1.7 years (1.1-3-1) in the 1-dose arm and 1.8 years (1.4-2.5) in the 2-dose arm, respectively.
Figure 1a + b: a) serological response at week 24; b) Comparison of Anti-HAV IgG (S/CO value) titer between both groups.
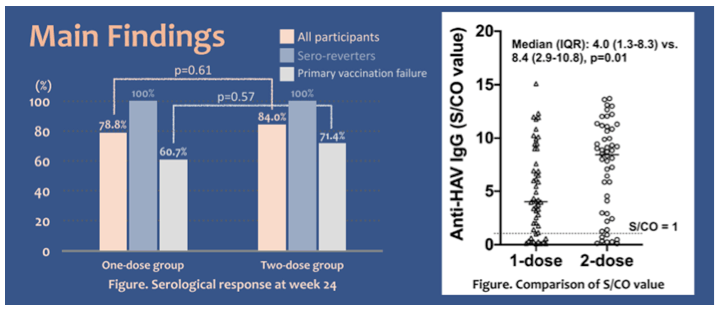
These findings clearly suggest that PLWH who develop no response to HAV vaccination or lose their antibodies over time can be successfully revaccinated with no significant difference between one dose or two dose revaccinations. PLWH who received 2 doses of HAV vaccine however, had higher anti-HAV IgG levels than those who received 1 dose of vaccine.
Hepatitis B
Chronic hepatitis B is a worldwide public health challenge. Knowledge of natural history of chronic hepatitis B is important for the management of the disease. In chronic HBV monoinfection HBV DNA levels are strongly associated with risk for HCC development (19).
Although HBV coinfection is common in HIV, the determinants of HCC in HIV/HBV coinfection are less well characterized. In the CROI oral abstract session on coinfections a study was presented which examined the incidence and predictors of HCC in a multi-cohort study of HIV/HBV-coinfected individuals (2). All HIV/HBV-coinfected persons (defined by positive HBV surface-antigen, HBe-antigen, or HBV DNA) within 22 cohorts of the North American AIDS Cohort Collaboration on Research and Design from 1995-2016 were included into the study. The main outcome, first occurrence of HCC, was verified by medical record review and/or cancer registry. Multivariable Cox regression was used to determine adjusted hazard ratios (aHRs [95% confidence intervals]) of factors assessed at cohort entry (age, sex, race, body mass index), ever during observation (hepatitis C coinfection, heavy alcohol use defined as alcohol use disorder diagnosis and/ or ≥3 drinks/day or ≥7 drinks/week for females; ≥4 drinks/day or ≥14 drinks/ week for males on AUDIT-C), or that were time-updated (HIV RNA, CD4+ cell percentage, diabetes mellitus, HBV DNA, HBV-active antiretroviral therapy [ART]). Figure 2 summarizes the selection criteria and the composition of the study participants.
Figure 2: Study flow of participants and selection criteria

ALT or AST >1,000 U/L within ±30 days of the first positive HBV laboratory test
Among 8,354 HIV/HBV-coinfected individuals (median age, 43 years; 93% male; 52.4% non-white), 115 HCC cases were diagnosed over 65,392 person-years of follow-up (incidence rate, 1.8 [95% CI, 1.5-2.1] events/1,000 person-years). The results for traditional HCC risk factors are summarized in table3.
Table 3: Traditional risk factors for HCC
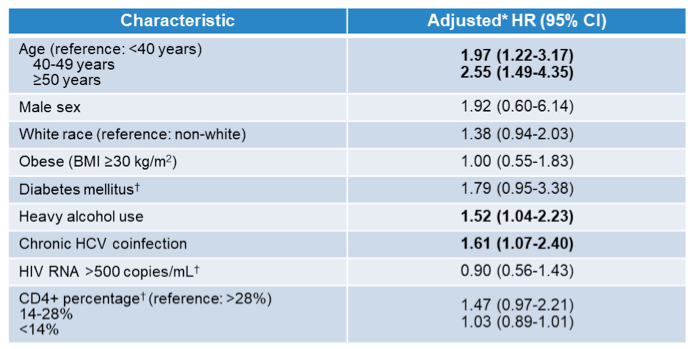
Within this analysis age was the most important risk factor for development of HCC followed by heavy alcohol use and concomitant HCV coinfection. The analysis evaluating the impact of HBV-DNA level is summarized in table 4.
Table 4: Risk of HCC by Quantitative HBV DNA
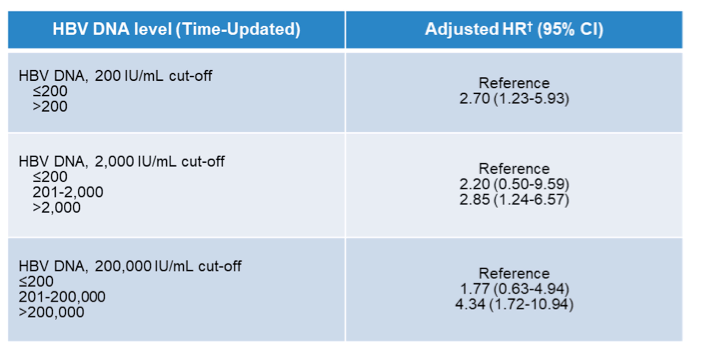
Among the 3,054 who had HBV DNA quantified, the risk of HCC was increased with HBV DNA >200 IU/mL (aHR, 2.70 [1.23-5.93]) and especially at levels >200,000 IU/mL (aHR, 4.34 [1.72-10.94], see Table). HCC risk was also higher with each 1.0 log10 IU/mL increase in HBV DNA (aHR, 1.18 [1.05-1.34]). HBV suppression <200 IU/ml with HBV-active ART for ≥1 year significantly reduced the risk of HCC (aHR, 0.42 [0.24-0.73]).
These findings highlight the independently increased risk for HCC with increasing HBV-DNA levels. This is meaningful particularly in HIV/HBV coinfection where time to achieving suppressed HBV-DNA levels under HBV therapy can be longer as in HBV monoinfection, as HIV/HBV coinfection is characterized by especially high baseline HBV viral loads. It poses the questions whether additional efforts should be undertaken to achieve complete HBV suppression as soon as possible which however, with the current HBV treatment options can be quite challenging.
In this context an interesting study from the Swiss Cohort Study was presented which aimed at describing the proportion of PLWH with persistent HBV replication despite tenofovir and to identify associated risk factors (7). PLWH coinfected with HBV defined as two positive hepatitis B surface antigen (HBsAg) tests more than 6 month apart, with at least four years of TDF therapy and a positive HBsAg at TDF start were included into the study. Patients who had an HIV RNA >200 cp/ml at two years were considered to have suboptimal adherence to ART and were therefore excluded from the analyses. The authors determined the proportion of patients with persistent hepatitis B viremia (HBV DNA ≥20 IU/ml) after two years and at the latest follow-up, and explored related risk factors using multivariable logistic regression adjusted for gender, age, ethnicity, CDC Stage 3, CD4 count at TDF start, HBV DNA levels at TDF start, previous HBV treatment, and hepatitis D coinfection. Overall, 222 subjects were included with a median age of 41 years (26-47), 80% male, mostly MSM with only 8% people who inject drugs and mainly (64%) from Europe. The median CD4-count at start of tenofovir was 166 cells/µl (61-270), 58% were 3Tc experienced and median duration of cART was 7.1 years (4.3-9.9). The viral hepatitis characteristics are listed below in table 5.
Table 5: Viral hepatitis characteristics

At TDF start, 187 (84%) had detectable HBV DNA, and 129 (58%) had high-level viremia (HBV DNA >2000 IU/ml). Persistent hepatitis B viremia was present in 61/222 (27%) at two years, and in 39/222 (18%) at the latest follow-up. At two years, 6/61 (10%) patients with persistent hepatitis B viremia had high-level viremia, and 5/39 (13%) at the latest follow-up. In multivariable analysis, persistent hepatitis B viremia at two years was associated with CDC Stage 3 (OR 2.80, CI 1.18-6.64), and high HBV DNA levels at TDF start (OR 1.23, CI 1.07-1.44), whereas it was less likely in individuals with age over 40 years (OR 0.42, CI 0.18-0.97), or with hepatitis D coinfection. The corresponding forest plot results are depicted in figure 3.
Figure 3: Forest plot of possible risk factors for persistant hepatitis B viremia two years after starting tenofovir based cART (mulzivariate analysis).
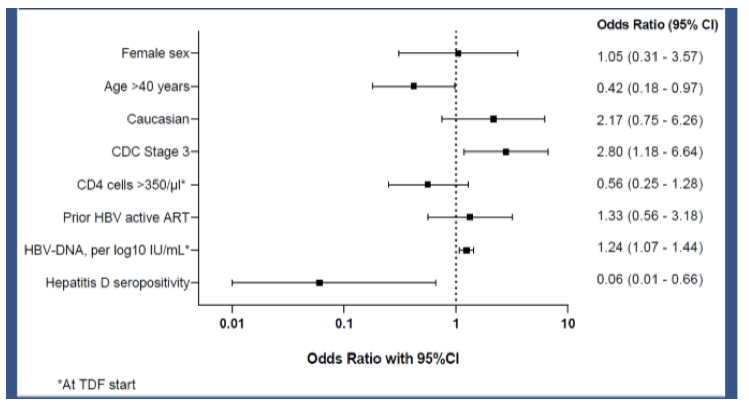
*adjusted for age, gender, ethnicity, CDC stage 3, CD4 count at TDF start, HBV DNA levels at TDF start, previous HBV treatment, and hepatitis D coinfection.
In conclusion, persistent hepatitis B replication after two years of TDF is frequent and associated with HBV DNA levels at TDF start. Patients with more advanced HIV infection appear to have higher baseline HBV viremia which needs longer time to achieve HBV suppression.
Another interesting study on the long-term outcome of tenofovir based HBV therapy in HIV -coinfection was also presented from the Swiss Cohort Study (8). In patients with HIV who become HBV infected, the risk for developing chronic hepatitis B is 6 x times higher than in HBV monoinfection particularly with more advanced immunodeficiency. Therefore, the question has been whether with availability of cART patients could lose their HBsAg after achieving immune reconstitution. A recent study analysed data on determinants of HBsAg loss in a retrospective multicentric cohort of 359 HIV/HBV coinfected patients (20). After a median follow-up of 11 years HBsAg loss had occurred in 66/359 (18%) patients. However, patients with stage CDC C (P ≤ .001), lower CD4 gain (P = .043) and not receiving TDF/FTC (P = .008) were less likely to lose HBsAg. Long-term TDF-containing cART appears to achieve higher rates of HBsAg seroclearance compared to published data for HBV monoinfected subjects. A similar question was now asked within the Swiss Cohort study indeed confirming a high rate of HBsAg loss in this study as well. A total of 272 patients were included. Median age was 41 years (IQR 36-46) and 221 (81%) were men. At tenofovir start, 110 (49%) patients were hepatitis B envelope antigen (HBeAg) positive, 229 (84%) had detectable HBV-DNA (median 1050 IU/ml, IQR 89-1.1x10E6) and 21% had low qHBsAg. HBsAg loss was observed in 8% (19/230) of participants during the first 2 years of tenofovir-containing ART and in 16% (43/262) of them after a median follow-up time of 8.4 years (IQR 2.6-15.8). At the last follow up, 54% (16/27) of those with HBsAg loss seroconverted for Anti-HBs antibodies. In multivariable analysis, low qHbsAg at tenofovir start (OR 12.01, CI 2.50-57.71) as well as female gender (OR 9.15, CI 1.08-77.45) were significant predictors of HBsAg loss, whereas this outcome was less likely among participants with negative baseline
HBV DNA (OR 0.14, CI 0.02-0.79). These findings underline that in patients with chronic hepatitis B and HIV-coinfection regular monitoring of HBsAg (or HBsAg levels) on an annual basis can be helpful to determine HBsAg loss. This will become even more important when patients want to switch their oral cART regimen to other modes of delivery. Patients with HBsAg loss and development of anti-HBs-antibodies for more than 6 months can be safely switched to non-tenofovir containing regimens.
Approximately 257 million people are chronically infected with hepatitis B worldwide. The implementation of rigorous vaccination programs has led to an overall decrease in the prevalence of this disease globally but this may also have led to emergence of viral mutations that can escape the protection of hepatitis B surface antibody. As this phenomenon is increasingly recognized, concern for transmission to vaccinated individuals has also been raised. Limited information is available on the circulation of vaccine-escape mutations, over time and their impact on serological parameters in the setting of HBV genotype-D. A research group from Rome presented data at CROI (3). Within this study escape variants at 12 HBsAg major hydrophilic region positions were studied in HBsAg sequences from 947 viremic HBV genotype-D infected patients (pts) collected from 2005 to 2019. A complex mutational profile was defined as one with 2 or more escape mutations. Almost 1 in 5 participants, 17.8%, had 1 or more vaccine-escape mutations, 14.7% had just 1 escape mutation, and 3.1% had a complex mutational profile. Subgenotype D3 had a higher prevalence of vaccine-escape mutations (22.9%) than subgenotypes D9 (15.4%), D2 (14.7%), D7 (14.3%), or D1 (13.4%) (P < 0.001). The most frequent individual escape mutations were P120S (21.4%), T131N (15.5%), and D144E (13.1%). Notably, the proportion of patients with complex profiles of vaccine-escape mutations increased over time: from 0.4% (1/237) in 2005-2009 to 3.0% (12/396) in 2010-2014 and to 5.1% (16/314) in 2015-2019, P= 0.007, suggesting an increased circulation of viral strains with enhanced capability to evade humoral responses. Moreover, the presence of complex profiles correlates with lower HBsAg levels. In conclusion, complex profiles of vaccine-escape mutations are detected in an increasing fraction of HBV genotype-D infected pts, and correlate with lower HBsAg quantification and HBsAg-negativity despite ongoing viral replication. These mutations may impact HBsAg results and need to be considered when developing new vaccine formulations.
Hepatitis D
Hepatitis delta (also known as hepatitis D or HDV) is a liver infection caused by the hepatitis delta virus that results in the most severe form of viral hepatitis known to humans. Only those already infected with hepatitis B can acquire hepatitis delta, as it is dependent on the hepatitis B virus to reproduce. Interesting data for delta hepatitis coinfection was presented at this year CROI meeting (9,10). A study from Taiwan investigated the recent trends of incident HDV infection among PLWH who had chronic HBV infection at a University Hospital in Taiwan between 2011 and 2018 (9). HBV serologic markers were determined on entry into care and during follow-up. Sequentially archived blood samples collected from PLWH with chronic HBV infection were retrieved for determinations of anti-HDV antibodies. Blood samples were retrospectively tested for anti-HDV antibodies for those who tested seropositive for HDV during the follow-up. The timing of incident HDV infection was estimated by the midpoint of the last time-point for HDV-seronegative samples and the first timepoint for HDV-seropositive samples collected. Overall, of 4096 PLWH seeking HIV care at the Taiwan University Hospital during the study period, 505 (12.3%) PLWH who had chronic HBV infection were included after exclusion of those who had no HBV infection at baseline and those without sequential blood samples. Thirty-three (6.5%) PLWH tested seropositive for HDV at baseline. During the follow-up, 41 (8.1%) PLWH, all being male, seroconverted for HDV while on tenofovir-containing combination antiretroviral therapy, with a median age of 43 years (IQR, 35.5-47.7). Of these 41 HDV seroconverters, 36 (87.8%) were men who have sex with men, 2 (4.9%) injection drug users and 2 (4.9%) heterosexuals. Plasma HIV RNA was <20 copies/ml in 61.9% (27/41) and HBV DNA <20 IU/ml in 77.3% (17/22) of the HDV seroconverters. Syphilis was diagnosed in 15.4% of seroconverters, 12.8% tested HCV-seropositive, and 37.1% had persistently elevated aminotransferase levels. The incidence rates increased from 4.66 per 1000 person-years of follow-up (PYFU) in 2011 to 13.81 per 1000 PYFU in 2015. The incidence rate spiked in 2016, with a rate of 49.93 per 1000 PYFU, which fell to 13.80, 13.05, and 13.95 per 1000 PYFU, in 2017, 2018, and 2019, respectively. The study flow and HDV results are captured in figure 4.
Figure 4: Study flow and HDV characteristics.
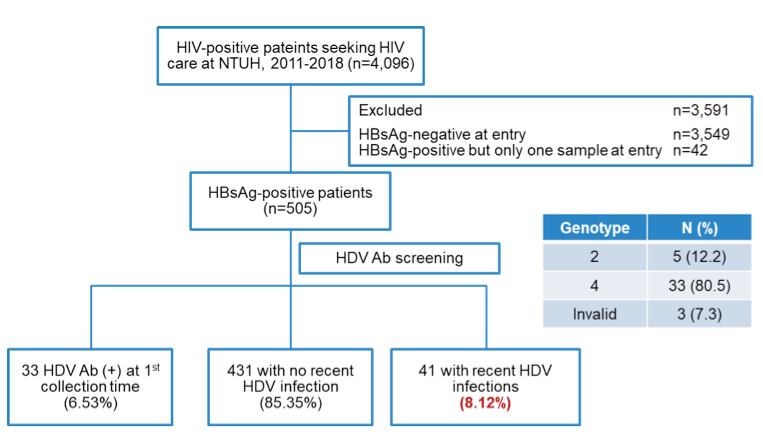
These results are important as they remind us of the ongoing risk for HDV superinfection in patients with chronic hepatis B. In most European cohorts IVDU are the main transmission group with increased risk for HDV infection. The high number of HDV infections in MSM within this study underlines the need for HDV screening also in this vulnerable population.
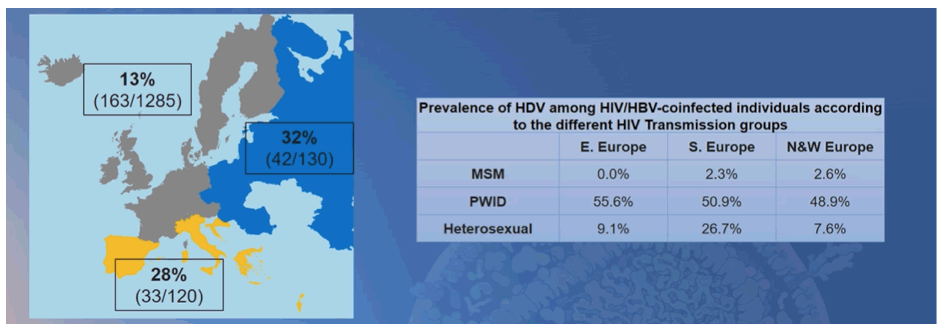
A European study analyzed data from EuroSIDA and the Swiss Cohort Study to characterize the current epidemiological trends in HDV infections across Europe (10). All PWH with a positive hepatitis B surface antigen (HBsAg) test were considered and tested for anti-HDV antibodies. HDV RNA amplification was performed in anti-HDV-positive patients. Demographic and clinical characteristics at initiation of antiretroviral therapy were compared between HDV-positive and HDV-negative individuals using descriptive statistics. The associations between HDV infection and overall mortality, liver-related mortality as well as hepatocellular carcinoma (HCC) were assessed using Kaplan-Meier and multivariable Cox regression adjusted for age, gender, HIV transmission group, baseline CD4 and cohort. Of 2793 HBsAg-positive patients, 1556 (56%) had stored serum available and were included into the analysis. The prevalence of HDV co-infection was 15.2% (237/1556, 95% CI: 13.5%-17.1%), of whom 66% (132/200) had active HDV replication. Figure 5 shows the HDV prevalence by European region and according to HIV transmission group.
Figure 5: Prevalence of HDV by region and HIV transmission risk group.
During a median follow-up time of 9.8 years [IQR 4-4-16.6], seventy-five (31.6%) HDV-positive patients and 261 (19.8%) HDV-negative individuals died. 43% (32/75) of the deaths were liver related in HDV-positive patients compared to 18% (46/261) in HDV-negative individuals. HDV infection was associated with overall mortality (adjusted hazard ratio 1.4; 95% CI 1.1-1.95, p=0.03), liver-related death (2.9, 1.6-5.1, p<0.001) and hepatocellular carcinoma (6.5, 2.6-16.6, p<0.001). These data strongly emphasize the negative impact of HDV coinfection on overall survival and risk for dying from liver disease related complications.
Hepatitis C
On the path to HCV elimination various barriers and hurdles in diagnosis and treatment of HCV have been identified. Costly pretreatment diagnostics and monitoring during HCV therapy are among those important barriers. The ACTG A5360 Study is a single-arm, open-label trial investigating the minimal monitoring (MINMON) approach for HCV treatment with sofosbuvir/velpatasvir for 12 weeks (1). A broad population of 399 subjects from 5 countries (United States (n=131), Brazil (n=131), Thailand (n=110), South Africa (n=15) and Uganda (n=12)) were treated. The main simplified study procedures are summarized in figure 6. The MINMON approach included: (1) no genotyping; (2) all tablets dispensed at entry; (3) no on-treatment visits/labs; and (4) two remote contacts at Weeks 4 (adherence assessment) and 22 (scheduling sustained virology response [SVR] visit). Unplanned visits for participant concerns (related/unrelated to an adverse event [AE]) were allowed. Median age was 47 years, 138 (35%) were cisgender women, 22 (6%) self-identified across the transgender spectrum, and 166 (42%) were White. At entry, 34 (9%) had compensated cirrhosis (FIB-4 ≥3.25) and 166 (42%) had HIV co-infection. Remote contact was successful at Weeks 4 and 22 for 394 (99%) and 335 (84%) participants, respectively.
Figure 6: Simplified monitoring approach in the MINMON trial.
Overall, 95% (379/399) achieved SVR (95% CI: 92.4%, 96.7%) (see figure 7).
Figure 7: SVR12 results.
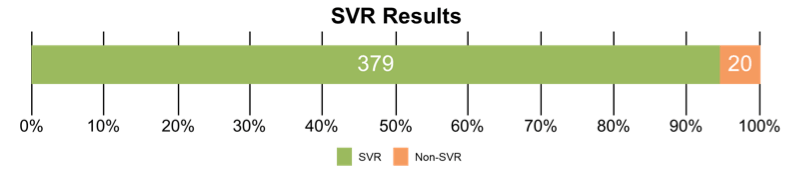
These results demonstrate clearly that less monitoring does not lead to more treatment failures which is extremely reassuring and may help to decrease barriers in getting access to HCV therapy. Wider adoption of this approach coupled with innovative case-finding strategies may contribute to HCV elimination while minimizing in-person appointments and resource use.
Restricting access to DAA therapy based on alcohol use is an additional barrier to DAA therapy and challenges HCV elimination goals. At this year CROI data was presented which examined the relationship between alcohol use, assessed within the 18 months prior to DAA therapy, and SVR (11). Using the Veterans Health Administration (VA) Birth Cohort, which includes all Veterans born between 1945 and 1965, all HCV patients who dispensed DAA therapy between 1 January 2014 and 31 December 2018 were identified. Alcohol use (abstinent, abstinent with history of alcohol use disorder [AUD], lower risk, moderate risk, high risk) was determined using a combination of self-reported score from the Alcohol Use Disorders Identification Test-Consumption instrument and validated International Classification of Diseases, 9th/10th Revision AUD diagnoses. Overall, 77045 HCV infected individuals dispensed DAA HCV therapy during the study period. Mean age was 63 years, 97% were male, 50% white, 40% black and 5% Hispanic. The majority of them with 84% had a genotype 1 HCV infection and only 3% were also HIV-coinfected. An impressive 94% of all study subjects achieved SVR defined as undetectable HCV RNA ≥12 weeks after DAA therapy. Alcohol use prior to DAA therapy varied: abstinent (47%), abstinent with AUD (13%) lower risk (19%), moderate risk (5%), and high risk (17%). Multivariable logistic regression was used to determine odds ratios (ORs) with 95% confidence intervals (CIs) of SVR for each alcohol use category compared to lower risk, adjusting for sex, age, residence type, BMI, DAA regimen, treatment year, FIB-4 score, liver cancer, smoking, HCV genotype, HBV coinfection, and hepatic decompensation. The results of the logistic regression analysis by alcohol use category are summarized in table 6.
Table 6: Adjusted Odds Ratio by alcohol use category
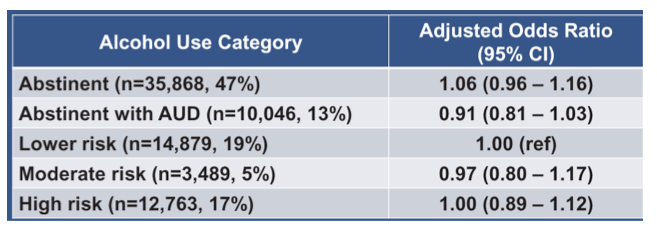
In summary, level of alcohol use, even at the highest category of use, was not associated with a lower likelihood of achieving SVR after adjusting for important differences between the groups. These findings suggest that DAA therapy should be provided and reimbursed despite ongoing alcohol use. Restricting access to DAA therapy based on alcohol use is an unnecessary barrier to DAA therapy and challenges HCV elimination goals.
Another potential challenge for achieving HCV elimination is the high rate of reinfections particularly in HIV+ as well as HIV- MSM under PrEP therapy. At CROI data from the German NoCo cohort was presented which evaluated factors associated with HCV reinfection(s) among MSM in Germany (12). The German NoCo cohort consists of patients from six German HIV and hepatitis treatment sites providing care for more than 8000 HIV-positive MSM and serving as primary care providers for HIV- MSM and HIV pre-exposure prophylaxis (PrEP) sites. Virologic data, HCV treatment data, risk factors, and behavior as well as liver disease assessment are assessed regularly. In this analysis, patients who were diagnosed with recently acquired HCV reinfection since 2014 up to 2020 were evaluated as a subgroup and compared to patients with a single HCV infection. Overall, 222 HCV episodes were recorded in the NoCo cohort with almost 70% only having one HCV episode. The number of HCV infections per study subject is shown in figure 8. 15.8% of patients cleared their HCV infection spontaneously, 47.4% were successfully treated with DAAs, 33.3 % achieved SVR with PEG/RBV. The reinfection incidence rate was 19.01/100 Person years (CI 15.50-23.26).
Figure 8: Number of HCV episodes per case (N=222) 2014 -2020
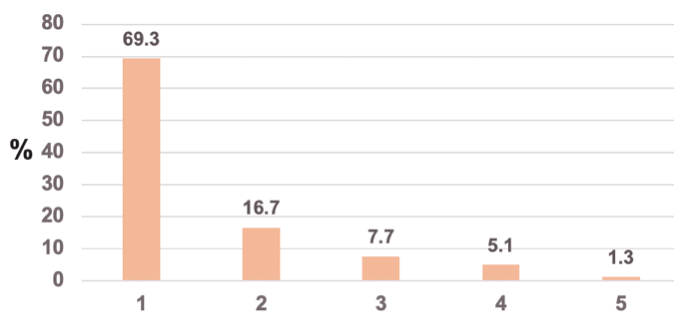
Compared to patients with primary HCV infection, reinfection cases were older, more often coinfected with HIV, and declared more often the use of crystal methamphetamine and had a trend towards declared ketamine use. Table 7 summarizes the main findings from the univariate analysis. Compared to patients with a single HCV reinfection, patients with multiple reinfections were not significantly different with regards to demographics, STD history, mode of HCV transmission, or substance use.
Table 7: Results: Univariate analysis
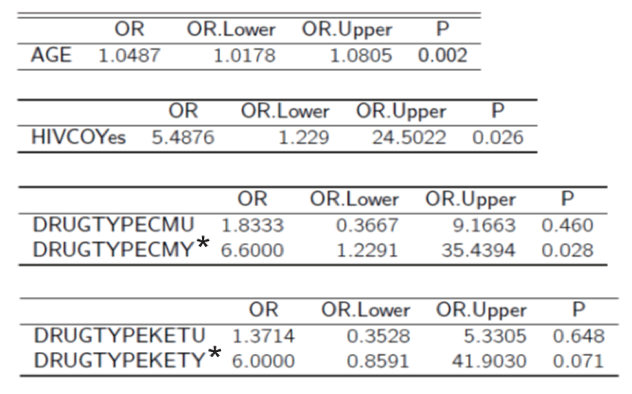
*CMY: crystal methamphetamine; KETY: ketamine
In the German NoCo cohort, HCV reinfection is frequent, especially in an aging HIV coinfected population with methamphetamine use. The role of recreational ketamine in this setting needs further study, as well as the (so far) infrequent detection of HCV reinfection in the HIV-negative MSM population. No specific behavior pattern could be identified for patients with multiple HCV reinfections.
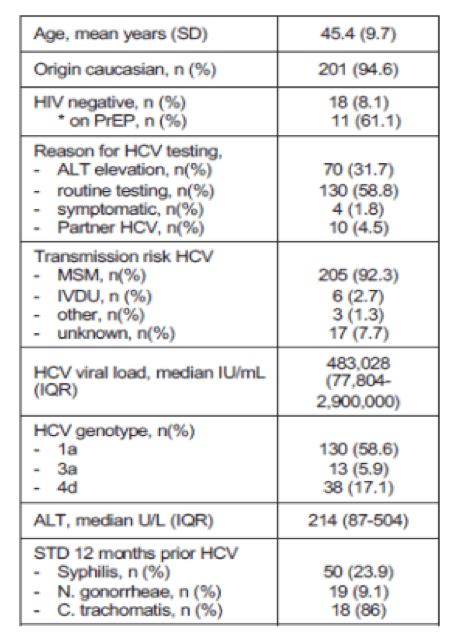
A second presentation from the NoCo cohort assessed the incidence of recently acquired HCV infection among MSM in Germany as well as HCV treatment uptake since 2014 (13). Various other countries have already reported a decline in new incident HCV infections in MSM following the universal access to DAA therapy for treatment of recent HCV infection (21,22). The NoCo cohort enrolled patients who were diagnosed with recently acquired HCV infection (<12 months) or reinfection since 2014. Virological data, HCV treatment data, risk factors, and behavior as well as liver disease status are assessed on a 3-6 months basis. The baseline characteristics of 222 subjects with recently acquired HCV infection are shown in table 8.
Table 8: Baseline characteristics
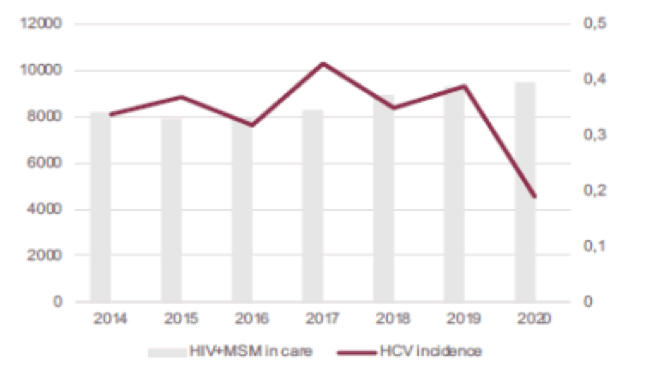
The incidence of recently acquired HCV infection over time is shown in figure 9. In this preliminary analysis from the German NoCo cohort, HCV incidence remained stable until 2019 despite a broad and successful use of DAAs. A drop in incidence in 2020 may be related to testing patterns, changes in care, or changes in behavior due to the SARS-CoV-2 pandemic.
Figure 9: Incidence of recently acquired HCV infection In HIV+ MSM
No DAA treatment failures have occurred in the NoCo cohort to date. The median time to initiation of HCV treatment was 6.6 months (IQR 3.9-9.7). In 36 patients (18%), HCV treatment was not initiated. Reason for no HCV treatment was Patient´s decision n=13 (36%), no coverage by health insurance n=12 (33%) or postponed but planned n=9 (25%). Delays to HCV treatment initiation and health insurance constraints may be contributing factors to ongoing HCV transmission in MSM in Germany.
There also was a study from Southern Spain at virtual CROI 2021 which estimated the incidence of and the factors associated with recently acquired HCV infection (RAHC) among patients sexually infected by HIV (14). Although, the incidence of recently acquired hepatitis C among HIV patients decreased after 2016, a lower but steady incidence of residual HCV cases remained from there on. New infections were associated with sharing chem-sex drugs among MSM. The authors postulated that early detection by intensified screening and immediate treatment of HIV-uninfected MSM with HCV infection to access potential reservoirs are needed to keep Spain on track for HV elimination by 2030.
Clearly, improved testing strategies for the high-risk populations for acquiring HCV infection such as MSM are needed. At virtual CROI data from a cost-effectiveness model were presented (15). The researchers hypothesized that better testing could result in a lower rate of transmission. They therefore, used a cost-effectiveness model to determine the optimal HCV testing strategies for HCV elimination among MSM. The model assumed 15% HIV prevalence among MSM, 8% HCV prevalence among MSM with HIV, and 25% PrEP coverage among MSM without HIV. Testing strategies were evaluated which could achieve a 90% reduction in HCV incidence from 2015 as a baseline, through 2030. The following HCV case-finding strategies were assessed: screening in parallel with HIV testing in PrEP non-users; screening every 12/6/3 months in MSM using PrEP; and screening every 6 months in
MSM with HIV. These strategies were considered alone and in combination, with the cost-effectiveness compared incrementally. Costs (USD) and quality adjusted life-years (QALYs) were assessed to estimate the mean incremental cost-effectiveness ratio (ICER) through 2050, compared to a willingness-to-pay (WTP) threshold of $100,000/QALY gained. The economic model predicted the optimal HCV testing strategy to achieve HCV elimination among MSM in the US to be every 6 months for MSM with HIV and annually for MSM using PrEP. The authors concluded that HCV elimination can be achieved among MSM in the US by a relatively nominal, logistically feasible, and cost-effective increase in the frequency of HCV screening over what is currently recommended by CDC and IDSA/AASLD. Indeed, EACS guidelines version 10.1 even recommends shorter screening intervals for PLWH and persistent HCV transmission risk: PLWH engaging in activities associated with increased risk of HCV transmission(iv) should be tested for HCV infection every 3 to 6 months.
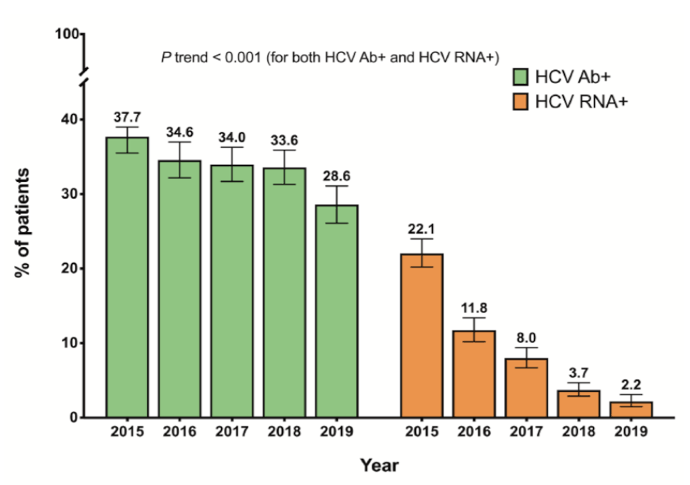
Microelimination of HCV in HIV/HCV coinfected individuals can be considered a low hanging fruit. Clearly, EACS guidelines recommend HCV screening in every HIV-infected person and HCV treatment in case chronic HCV is diagnosed. Overall cure rates are above 95% with the nowadays available modern pangenotypic all oral DAA regimens after 8-12 weeks of therapy. So eventually, prevalence of active HCV infections should decline dramatically on a national level. Very nice data was presented from Spain assessing the prevalence of HCV-antibodies and active HCV infection (HCV RNA +) in people living with HIV in Spain in 2019 and compared to results with four similar studies performed yearly from 2015 to 2018 (16). All oral DAA-based therapy has been available in Spain since the third trimester of 2014. Since June 2017, free access to treatment has been available to all HCV-infected individuals. The study was performed in 41 centers (October-November, 2019) with a reference population comprised of 41,973 PLWH. A sample size was determined by proportional allocation, and patients were selected using simple random sampling (n=1325). HCV serostatus was known in 1,316 (99.3%), and 376 (28.6%) were HCV antibody-positive (78.7% PWID and 7.7% MSM). Of the 376 HCV antibody positive patients, 291 cleared HCV after anti-HCV therapy, 55 cleared HCV spontaneously, 29 were HCV-RNA-positive, and 1 had unknown HCV-RNA. The prevalence of HCV-RNA-positive was, therefore, 2.2%. As 11 of 29 patients were receiving DAAs, and assuming treatment effectiveness of 95%, the HCV-RNA positive prevalence could be considered to be 1.4%. Reasons for not receiving anti-HCV therapy in 18 patients included physician decision (N=5), loss to follow-up (N=3), patient refusal (N=2), and unknown reasons (N=4). The prevalence for HCV antibodies as well for HCV-RNA positive patients over time is depicted in figure 10. The prevalence of HCV Ab decreased steadily from 37.7% in 2015 to 28.6% in 2019 (P <.001). Likewise, HCV-RNA prevalence decreased from 22.1% in 2015 to 2.2% in 2019 (P <.001).
Figure 10: HIV/HCV Coinfection trends in Spain (2015-2019): Prevalence of HCV
Treatment uptake over time in the Spanish cohort is shown in figure 11. Anti-HCV treatment uptake increased from 53.9% in 2015 to 95.0% in 2019 (p<.001).
Figure 11: HCV treatment uptake
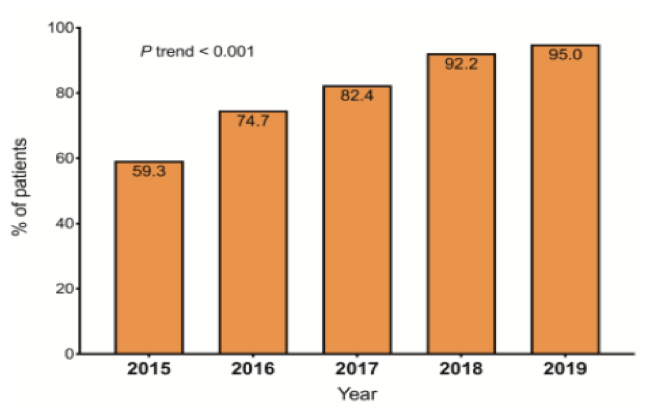
In conclusion, the elimination of HCV infection among PLWH in Spain is an achievable goal shortly.
HIV and fatty liver disease
Non-alcoholic fatty liver disease (NAFLD) is becoming a serious complication for people living with HIV (PLWH) - reported prevalence ranges from 13-73% (23,24). Transient elastography (TE) and controlled attenuation parameter (CAP) are validated and widely used as non-invasive screening tools in clinical practice but there is also an ongoing search for new diagnostic biomarkers and screening tools. More recently, a novel FibroScan-AST (FAST) score had been validated in HIV negative individuals with suspected NAFLD (25). The corresponding formula is shown below in figure 12. The aim of this study was to evaluate the new FibroScan-AST Score in an HIV monoinfected cohort (17).
Figure 12: FAST score

Overall, 432 HIV positive participants presenting at the Outpatient HIV Clinic at University Hospital Bonn, Germany were enrolled. Enrolment started on 1st of August 2013 and annual follow-up is still ongoing. For this analysis all visits until 31th of December 2018 were included. For this analysis 319 HIV+ monoinfected participants with ≥ 2 visits included. 77% of the study population were male, 76% Caucasian, 95% under cART, 86% virally suppressed and 6% had diabetes mellitus. The distribution of the FAST score over the study period is captured in figure 13, the distribution of CAP values in figure 14.
Figure 13: Distribution of FAST score over the study period 2013-2018
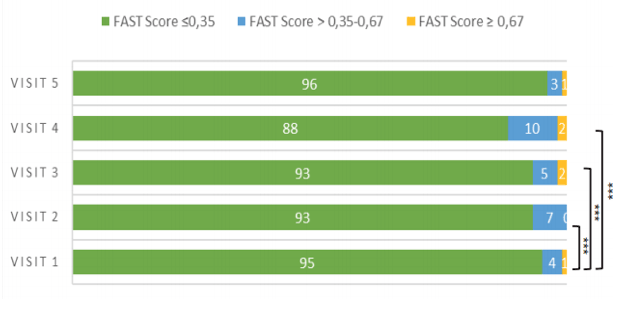
** p<0.01 *** p<0.001
Figure 14: Distribution of CAP values over the study period
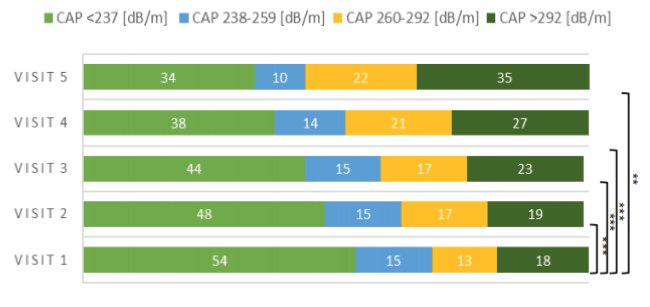
In contrast to an observed steady redistribution towards higher CAP values (reflecting an increase in steatosis) over five years, the FAST score showed less changes in its distribution over the study period. However, at least within the first four years with a larger n of study subjects there was a slight increase in more pronounced steatosis stages over time. Higher FAST Scores in univariate analysis were associated with male sex, diabetes mellitus 2 and fasting glucose, obesity (BMI >25kg/m2), cholesterol, yGT and ever having received an integrase inhibitor. All parameters however, lost significance in multivariable analysis. FAST score at baseline showed a strong correlation with baseline FIB4 scores (R:0,529; p<0.001) and APRI scores (R: 0,758; p<0.001) but was weaker with CAP values (R: 0,371; p<0.001).
In conclusion, the authors found a significant correlation between FAST-score and FIB4, APRI and CAP values. The FAST score should be further evaluated in HIV-patients.
At CROI there was also a further study presented which aimed at validating non-invasive markers for the diagnosis of NAFLD in HIV-infected patients (18). With the increasing prevalence of NAFLD in HIV and an overall aging patient population with increasing risks for non-AIDS defining comorbidities this is of great interest. This prospective cohort study in Spain included HIV-infected subjects on stable ART regimen and persistent liver enzymes elevation without known liver disease. Patients underwent whole blood lab test, abdominal ultrasound (US), transient elastography (including CAP) as well as assessment of non-invasive steatosis and fibrosis markers (TyG, HSI, FLI, FIB-4 and APRI). In addition, a liver biopsy was offered to all patients and performed in those who consented. AUROC analysis was performed to estimate the diagnostic accuracy of non-invasive tests of both steatosis and fibrosis compared to liver biopsy. An algorithm with serial combination of tests was developed. Overall 146 patients were enrolled of whom 69 accepted to undergo liver biopsy. Baseline characteristics of the total study population were as follows: 91% men, CDC C3 stage 14.5%, HIV RNA < 50 cop/mL 100%, median (IQR) age 49 years (41-54), BMI 29 (25-31), baseline CD4+ 829 cel/μL (593-930). Metabolic syndrome was diagnosed in 42% and diabetes mellitus or impaired fasting glucose in 45%. Medium values for transaminases were: ALT 57 IU/L (43-83), AST 37 IU/L (29-47), GGT 50 IU/L (31-118). In the biopsies 90% of patients had steatosis, 61% steatohepatitis, 30% had any grade of fibrosis and 4% ≥ F3 fibrosis. AUROC for US, CAP, FLI, HSI, TyG were: 0.90, 0.94, 0.81, 0.74 and 0.75 (p<0.05);
regarding liver fibrosis TE, APRI and FIB-4 were 0.95, 0.92 and 0.97 (p<0.05). Two non-invasive-combination models of tests with TyG and FLI or HSI as first tests and US or CAP as second tests had the best diagnosis performance for liver steatosis, AUROC 0.99 (0.97-1 p<0.001), 0.92 (0.77-1 p<0.001). The corresponding findings are shown in figure 15.
Figure 15: Non-invasive algorithm for diagnosis of NAFLD.
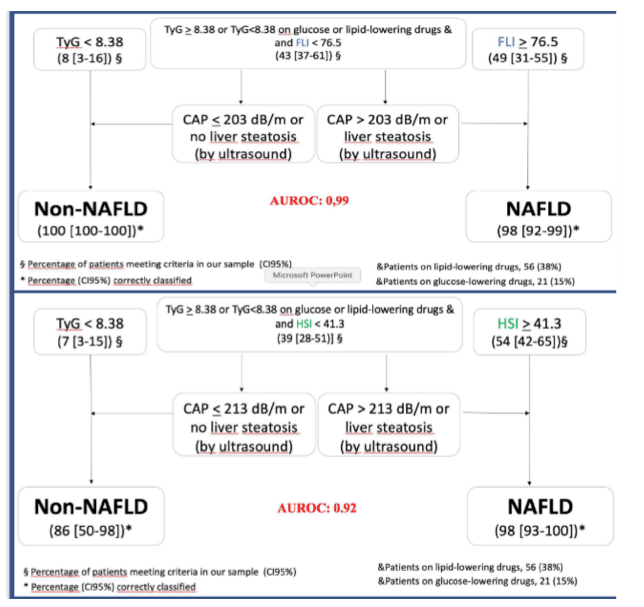
In summary, the combination of TyG with FLI or HSI as first tests and US or CAP as second tests is able to accurately diagnose or exclude the presence of NAFLD. This could help avert the need for liver biopsy for diagnosis of fatty liver disease in HIV. More data is urgently needed to come up with a non-invasive gold standard for NAFLD and NASH diagnosis.
Summary
• Hepatitis A vaccination rates in HIV coinfected patients are low and need to be improved.
• Among PLWH who have no responses to primary 2-dose HAV vaccination or those who’s anti-HAV antibodies have waned, revaccination with 1 or 2 doses of HAV vaccine result in similar serologic responses.
• HIV/HBV-coinfected individuals on ART with detectable HBV DNA >200 IU/mL remain at increased risk for HCC. Heavy alcohol use and hepatitis C coinfection are further important determinants of HCC.
• Persistent hepatitis B replication after two years of TDF is frequent and associated with HBV DNA levels at TDF start.
• Up to 16% of PLWH coinfected with HBV on tenofovir-containing ART lose their HBsAg over time. Interestingly, baseline quantitative HBsAg level is a strong predictor of this outcome.
• Complex profiles of vaccine-escape mutations are detected in a not negligible fraction of HBV genotype-D infected patients, and need to be taken into account for the development of novel vaccine formulations.
• HDV superinfections continue to occur among PLWH who have chronic HBV infection in the era of tenofovir-containing combination antiretroviral therapy. Every individual with chronic hepatitis B should be screened for hepatitis delta superinfection.
• Hepatitis delta prevalence among PWH in Europe varies strongly across regions and is particularly high in PWIDs. This could reflect different availability of harm reduction programs. HDV coinfection is associated with increased mortality and liver related events including HCC.
• Simplified HCV treatment with SOF/VEL over 12 weeks with no lab monitoring or visits leads to 95% SVR rate and may help to increase rollout of HCV therapy and contribute to HCV elimination.
• Level of alcohol use was demonstrated not to be associated with a lower likelihood of achieving SVR. Therefore, DAA therapy should be provided and reimbursed despite alcohol use. Restricting access to DAA therapy based on alcohol use is an unnecessary barrier to DAA therapy and challenges HCV elimination goals.
• HCV reinfection is frequent, especially in an aging HIV coinfected MSM population with methamphetamine use.
• Delays to HCV treatment initiation and health insurance constraints may be contributing factors to ongoing HCV transmission in MSM in Germany.
• The optimal cost-effective HCV testing strategy to achieve HCV elimination among MSM in the US is to screen for HCV every 6 months for MSM with HIV and annually for MSM using PrEP.
• The elimination of HCV infection among PLWH in Spain is an achievable goal.
• FAST Score is a rapidly gathered, simple and inexpensive screening tool for NASH in PLWH.
• The combination of TyG with FLI or HSI as first tests and US or CAP as second tests is able to accurately diagnose or exclude the presence of NAFLD. This could help avert the need for liver biopsy for diagnosis of fatty liver disease in HIV.
References
1. Solomon SS et al. A simple and safe approach to HCV treatment: Findings from the A5360 (MINMON) trial. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2020, abstract 135
2. Kim HN et al. Hepatocellular carcinoma and HBV viremia in HIV/HBV coinfected persons in NA-ACCORD. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2020, abstract 136
3. Plermatteo L et al. Uprising circulation of HBV-complex profiles with HBsAG vaccine-escape mutations. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2020, abstract 137
4. Ciocca ET et al. Retrospective study of hepatitis A immunization rates in persons with HIV. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2021, abstract 464
5. Fernandez-Fuertes M, et al. Low rate of vaccination and risk of incident hepatitis A among HIV-infected MSM. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2020, abstract 466
6. Chen GJ et al. HAV revaccination in seronegative or seroreverted PLWH after primary vaccination. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2021, abstract 465
7. Hofmann E et al. HBV replication during tenofovir therapy is frequent in HIV/HBV coinfection. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2020, abstract 450
8. Beguelin C et al. Frequent HBsAg clearance during tenofovir therapy in HIV/HBV coinfection. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2020, abstract 449
9. Hung CC et al. Recent trends of hepatitis D virus infection among people living with HIV in Taiwan. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2020, abstract 463
10. Beguelin C et al. Hepatitis delta infections among persons with HIV in Europe. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2020, abstract 452
11. Cartwright EJ et al. Relationship between alcohol use and sustained virological response to HCV DAA therapy. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2020, abstract 454
12. Boesecke C et al. Reinfection with the hepatitis B virus in men who hacve sex with men. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2020, abstract 439
13. Ingiliz P et al. No change in incidence of recently acquired HCV in HIV+ MSM in Germany (NOCO Cohort). 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2021, abstract 442
14. Gonzalez-Serna A et al. Incidence of recently acquired hepatitis C virus infection among HIV-infected patients in Southern Spain. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2021, abstract 445
15. Chaillon A et al. Cost-effectiveness of HCV testing strategies for HCV elimination among MSM in the US. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2021, abstract 440
16. Fanciulli C et al. HIV/HCV coinfection trends in Spain (2015-2019). 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2021, abstract 441
17. Bischoff J et al. Application of the novel FIBROSCAN-AST score in an HIV monoinfected cohort. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2021, abstract 455
18. Busca C et al. A serial combination of steatosis noninvasive markers in HIV-moninfected subjects. 28th Virtual Conference on Retroviruses and Opportunistic Infections, March 06-10, 2021, abstract 460
19. Chen CJ, et al.; REVEAL-HBV Study Group. Risk of hepatocellular carcinoma across a biological gradient of serum hepatitis B virus DNA level. JAMA. 2006;295:65-73.
20. Van Bremen K, et al. Obstacles to HBV functional cure: Late presentation in HIV and its impact on HBV seroconversion in HIV/HBV coinfection. Liver Int. 2020;40:2978-2981.
21. Boerekamps A, et al. Declining Hepatitis C Virus (HCV) Incidence in Dutch Human Immunodeficiency Virus-Positive Men Who Have Sex With Men After Unrestricted Access to HCV Therapy. Clin Infect Dis 2018;66:1360-1365.
22. Braun DL, et al. A treatment as prevention trial to eliminate hepatitis C among men who have sex with men living with HIV in the Swiss HIV Cohort Study. Clin Infect Dis. 2020 Aug 6:ciaa1124. doi: 10.1093/cid/ciaa1124. Epub ahead of print.
23. Sebastiani G, et al. Incidence and predictors of hepatic steatosis and fibrosis by serum biomarkers in a large cohort of human immunodeficiency virus mono-infected patients. Open Forum Infect Dis. 2015;2:ofv015.
24. Rockstroh JK. Non-Alcoholic Fatty Liver Disease (NAFLD) and Non-Alcoholic Steatohepatitis (NASH) in HIV. Curr HIV/AIDS Rep. 2017;14:47-53.
25. Newsome PN, et al. FibroScan-AST (FAST) score for the non-invasive identification of patients with non-alcoholic steatohepatitis with significant activity and fibrosis: a prospective derivation and global validation study. Lancet Gastroenterol Hepatol. 2020;5:362-373.
|
| |
|
 |
 |
|
|