 |
 |
 |
| |
NEXT-GENERATION ISLATRAVIR IMPLANTS PROJECTED TO PROVIDE YEARLY HIV PROPHYLAXIS
|
| |
| |
CROI 2021 March 6-10 Reported by Jules Levin
Randolph P . Matthews1, Xiaowei Zang1, Stephanie Barrett1, Adrian Goodey1, Tycho Heimbach1, Vanessa L. Weissler1, Carlien Leyssens1, Tom Reynders1, Ryan Vargo1, Yang Liu1, Robert Schwab2, Sylvie Rottey3, Michael N. Robertson1, Selwyn A. Stoch1, Marian Iwamoto1 1Merck & Co, Inc, Kenilworth, NJ, USA, 2Celerion, Lincoln, NE, USA, 3Ghent University Hospital, Ghent, Belgium
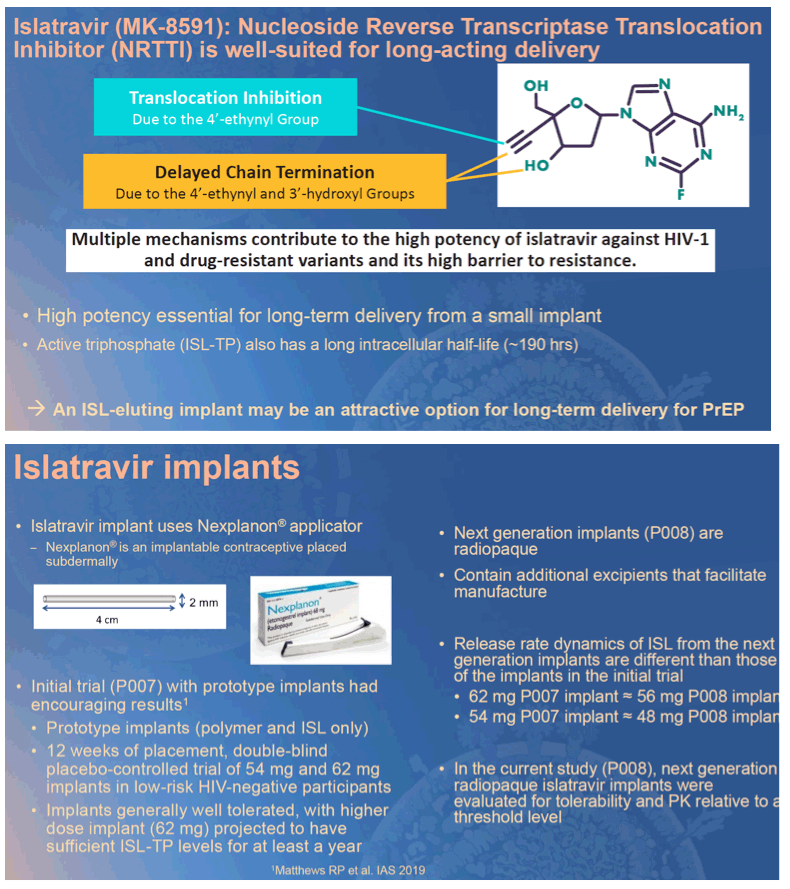
Background: Preexposure prophylaxis (PrEP) with antiretroviral drugs has demonstrated efficacy in reducing new HIV infections, although efficacy is tightly linked to good adherence, especially in women. Islatravir (MK-8591) is a nucleoside reverse transcriptase translocation inhibitor with high potency and long t1/2, currently in development for PrEP as an oral monthly pill. In addition, prototype islatravir implants (containing only polymer and islatravir) have demonstrated the potential for yearly administration for PrEP.
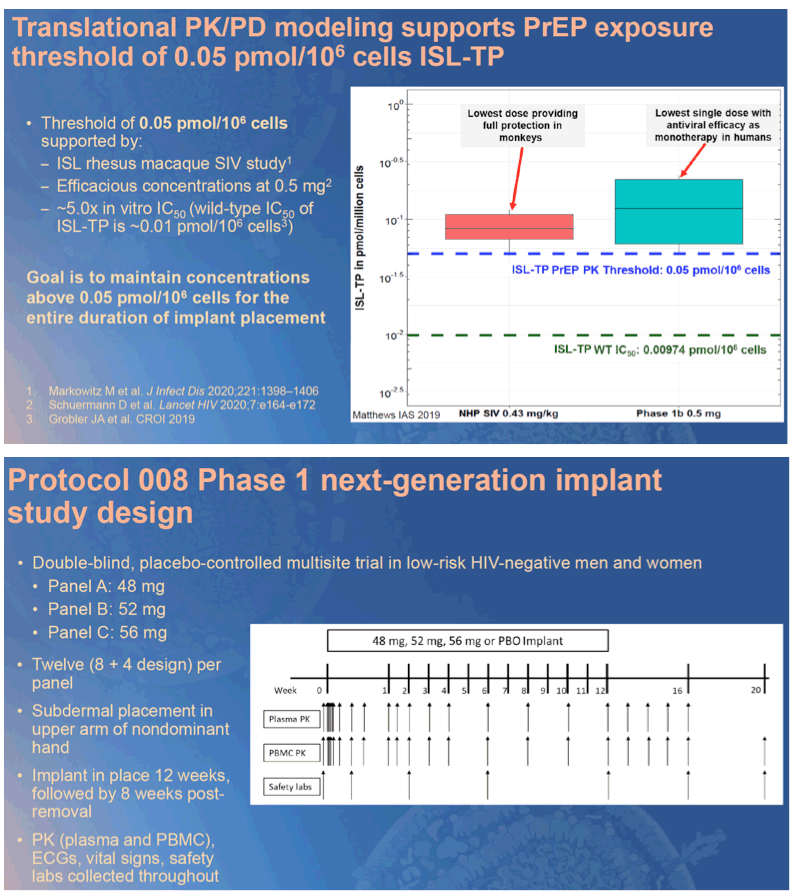
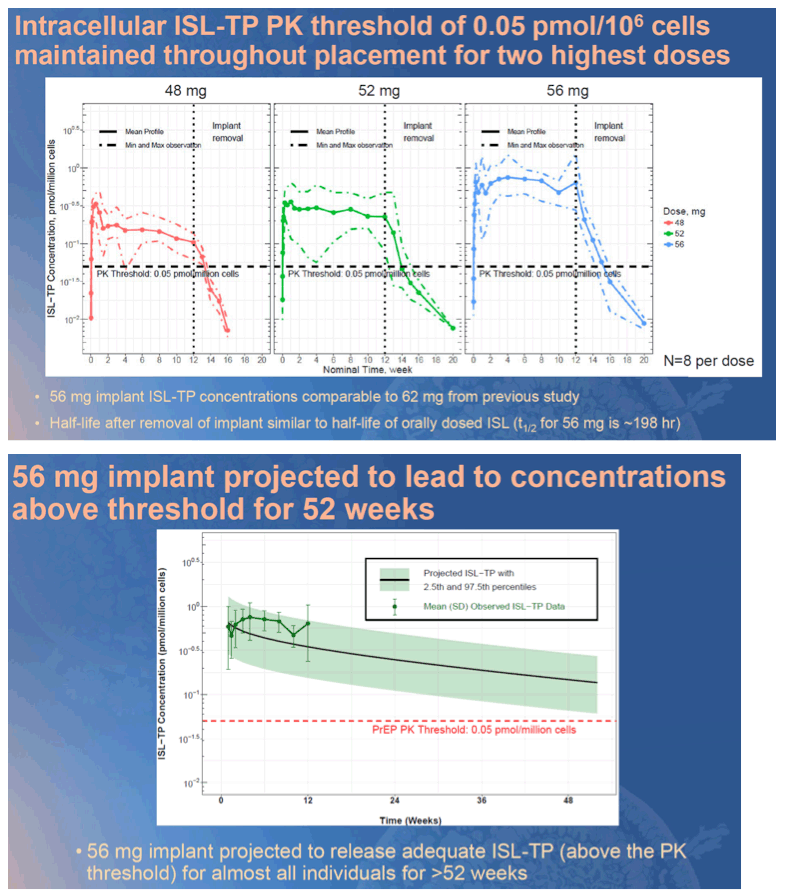
Methods: Radiopaque next-generation islatravir-eluting implants were studied preclinically to establish general tolerability and assess pharmacokinetics (PK) of islatravir parent and active islatravir-TP (triphosphate). These data, along with data from an SIV challenge study and from previous Phase 1 trials, formed the basis for establishing a threshold islatravir-TP concentration of 0.05 pmol/million cells in PBMCs. In this double-blind placebo- controlled multicenter Phase 1 trial, a single islatravir-eluting (48 mg, 52 mg or 56 mg) or placebo implant was placed in participants at low risk of HIV infection for 12 weeks. Safety and tolerability, as well as PK for islatravir parent and islatravir-TP from plasma and PBMCs, was collected throughout placement and for 8 weeks post removal.
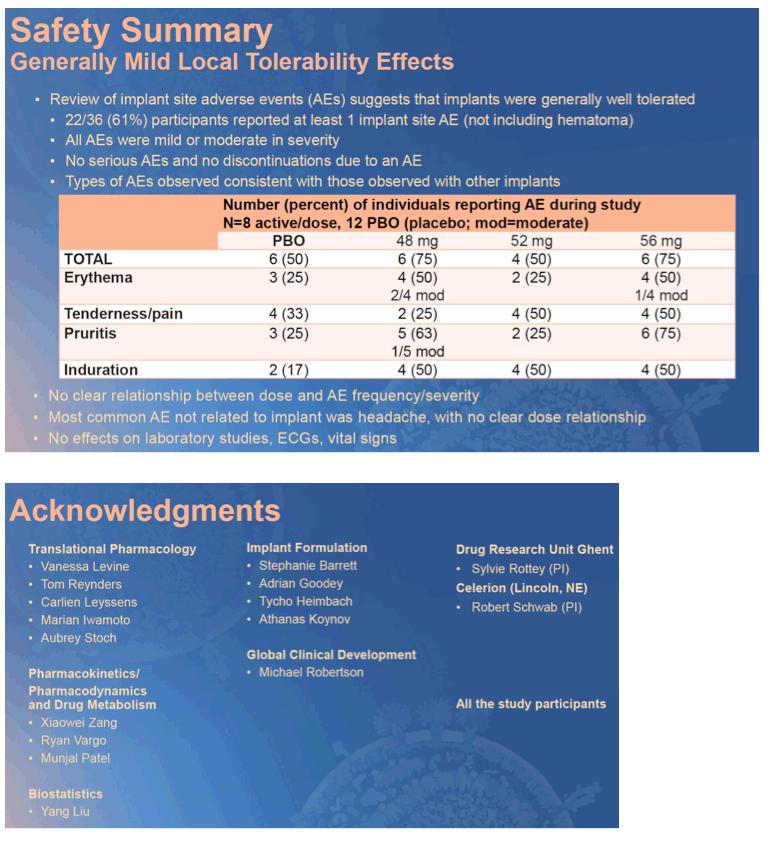
Results: Implants were generally well tolerated, and there was no clear dose-dependent difference in implant-related adverse events (Table 1A). Active islatravir-TP levels were above target for all implants throughout implant placement (Table 1B). Data from this trial and from in vitro assessments of the ISL implants suggest that implants of >52 mg will achieve mean ISL-TP concentrations above the PK threshold at 52 weeks.
Conclusion: Next-generation radiopaque islatravir-eluting implants provide drug release projected to be sufficient for HIV prophylaxis for at least one year. Islatravir-eluting implants appear to be well tolerated, and the results from this trial support further study of these implants in a larger, longer Phase 2 trial. A PrEP implant could provide an attractive option for individuals in whom adherence to a daily PrEP regimen is challenging.

|
| |
|
 |
 |
|
|