 |
 |
 |
| |
Integrase inhibitor drug resistance in clinical practice in the United Kingdom
|
| |
| |
K. El Bouzidi1,2, A. Tostevin1, A. Pozniak3, D. Asboe3, D. Churchill4, A. Phillips1, C. Orkin5, A. Winston6, C.A. Sabin1,7, D.T. Dunn1, on behalf of the UK CHIC Study and UK HIV Drug Resistance Database
1Institute for Global Health, University College London, London, United Kingdom, 2Division of Infection & Immunity, University College London, London, United Kingdom, 3Chelsea and Westminster Hospital NHS Foundation Trust, London, United Kingdom, 4Brighton and Sussex University Hospitals NHS Trust, Brighton, United Kingdom, 5Barts Health NHS Trust, London, United Kingdom, 6Department of Infectious Disease, Imperial College London, London, United Kingdom, 7National Institute for Health Research (NIHR) Health Protection Research Unit in Blood Borne and Sexually Transmitted Infections at University College London, London, United Kingdom
Purpose: Integrase strand transfer inhibitors (INSTIs) are now widely used to treat HIV. There is a need to understand the resistance patterns associated with virological failure on INSTI-containing regimens.
Method: HIV-1 clinical and sequence data from the UK Collaborative HIV Cohort (CHIC) study and UK HIV Drug Resistance Database were linked to investigate IAS-USA 2017 integrase resistance-associated mutations during INSTI-containing antiretroviral therapy. Results were stratified by current and previous INSTI agents.
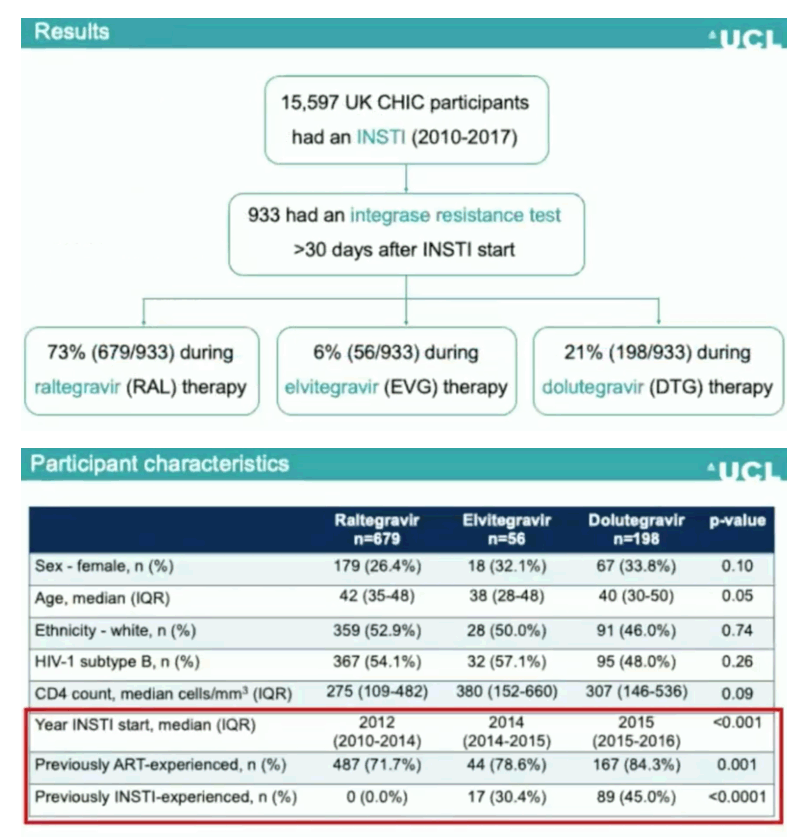
Results: 15,597 UK CHIC participants had received an INSTI up to 31/12/17. An integrase resistance test was performed during treatment failure for 933 participants: 71.7% men, median age 41 (interquartile range [IQR] 33-48), median baseline CD4 count 292 cells/mm3 (IQR 112-483), 52.9% subtype B and 27.8% ART-naive at the start of INSTI therapy. The INSTI prescribed at the time of resistance testing was raltegravir in 679 (72.8%), elvitegravir in 56 (6.0%), and dolutegravir in 198 (21.2%). When raltegravir and elvitegravir were used as the first INSTI agents, around 15% developed major integrase resistance during virological failure, compared with 0% for dolutegravir (Table). Overall, major integrase resistance mutations were detected during INSTI failure in 123 (13.2%) participants, mainly following raltegravir or elvitegravir use, including N155H (n=78), Q148H/K/R (n=24), Y143C/H/R (n=13), E92Q (n=7), T66I (n=3), F121Y (n=3), R263K (n=3) and G140R (n=1). Minor integrase mutations were present in 88 (9.4%) participants: T97A (n=51), L74M (n=17), E138A/K (n=14), G140A/S (n=13), T66A/K (n=5) and E92G (n=1).
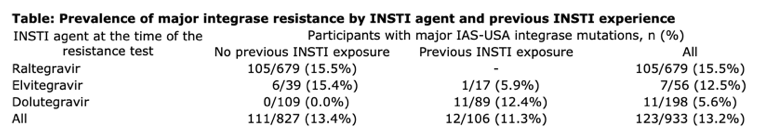
Conclusions: The emergence of major integrase resistance was fairly common in the era of mainly first-generation INSTI use. However, dolutegravir failure among people who were previously INSTI-naive was associated with a low risk of drug resistance. This may reflect changes in the patient populations exposed to INSTIs over time, as well as the higher genetic barrier to resistance afforded by dolutegravir.


|
| |
|
 |
 |
|
|