 |
 |
 |
| |
Short interfering RNA JNJ-3989 combination therapy
Sustained Drops in Four HBV Markers With siRNA Plus Nukes
|
| |
| |
EASL International Liver Congress, June 23-26, 2021 JNJ-3989
Mark Mascolini
When combined with nucleos(t)ide analogs (NAs), the short interfering RNA (siRNA) agent JNJ-3989 yielded sustained declines in four standard markers of HBV infection: hepatitis B surface antigen (HBsAg), hepatitis B e antigen (HBeAg), hepatitis B core-related antigen (HBcrAg), and HBV RNA-all of which offer measures of ongoing HBV infection or replication [1].
An international team of researchers (most in Australia and New Zealand) noted that NAs inhibit HBV replication but do not prevent HBsAg production. By targeting both the S gene and the X gene of HBV, the siRNA JNJ-3989 degrades all viral RNA and stifles production of all HBV RNA transcripts, such as HBsAg. Lowering HBsAg concentrations, the researchers explained, should restore the immune response in people with chronic HBV infection.
A phase 2b trial that combined JNJ-3989 with NAs in people with HBeAg-positive or -negative HBV infection found that treatment lowers levels of HBV markers (NCT03365947) [2]. This analysis of trial results had two objectives: (1) to assess the impact of baseline factors in lowering HBsAg during treatment and (2) to compare the effect of JNJ-3989 plus NAs on levels of HBsAg, HBeAg, HBcrAg, and HBV RNA.
Study participants could be HBeAg-positive or negative and NA-experienced or naive. All continued their NA (tenofovir disoproxil fumarate or entecavir) or started NA therapy when JNJ-3989 began. On days 0, 28, and 56, participants received JNJ-3989 at a subcutaneous dose of 100 mg (8 people), 200 mg (8 people), 300 mg (16 people), or 400 mg (8 people).
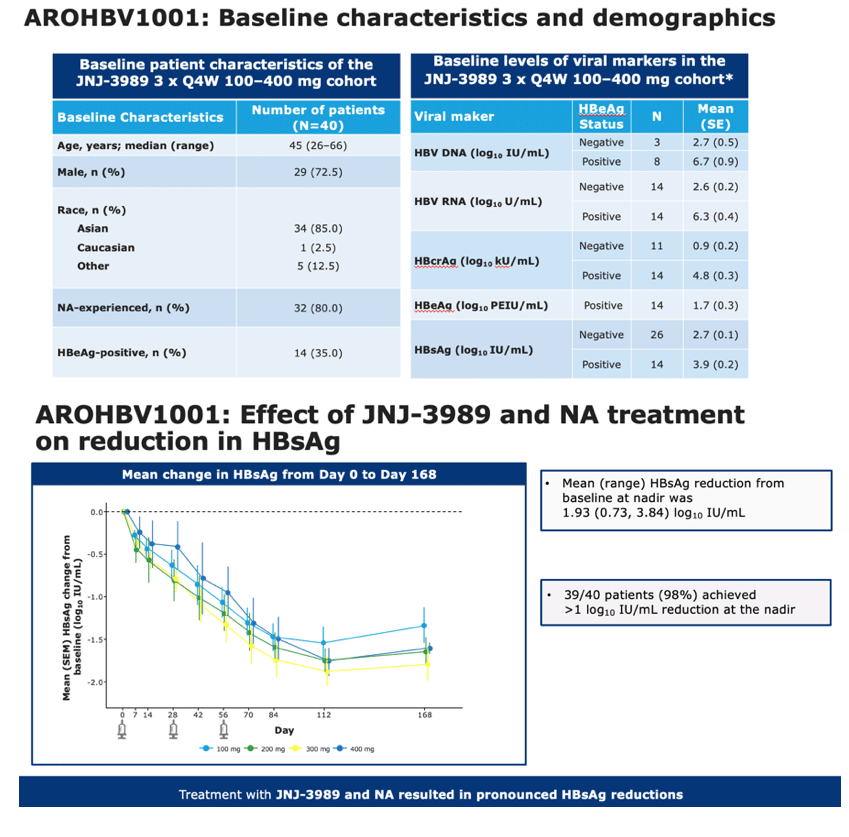
The 40 study participants had a median age of 45 years, 29 (72.5%) were men, 34 (85%) were Asian, 32 (80%) had NA experience, and 14 (35%) were HBeAg-positive.
By treatment day 168, average HBsAg fell 1.93 log10 IU/mL (range 0.73 to 3.84). Thirty-nine of the 40 participants (98%) had at least a 1 log10 (10-fold) HBsAg drop at the lowest point of the decline. While 28 people (70%) had less than a 2 log10 (100-fold) fall in HBsAg, 12 (30%) had a 2 log10 or greater reduction. When the trial began the under-2 log10 group differed from the over-2 log10 group in the following parameters:
- HBeAg-positive: 43% vs 57%
- HBeAg-negative: 85% vs 15%
- Female: 36% vs 64%
- Male: 83% vs 17%
- 40 years or younger: 40% vs 60%
- Older than 40: 88% vs 12%
Baseline levels of HBsAg, other viral markers, NA-naive vs NA-suppressed, and body mass index had no impact on the decrease in HBsAg during treatment. HBsAg tended to fall more on treatment in HBeAg-positive than negative people, but that outcome was largely driven by 4 HBeAg-positive women who had the biggest HBsAg declines (more than 2.5 log10 drop at the lowest point). HBV genotype had no impact on HBsAg decrease, but many participants did not have genotype data available.
HBeAg, HBcrAg, and HBV RNA also fell sharply during treatment, but HBeAg and HBcrAg declines tended to be smaller than drops in HBsAg. For those three viral markers-HBeAg, HBcrAg, and HBV RNA-the researchers compared declines at treatment day 113 (in log10) with HBsAg declines in people who had a baseline level more than 10-fold above the lower limit of detection for each of those three markers:
- HBeAg -1.47 (0.12 to 12) vs HBsAg -2.12 (0.21 to 12)
- HBcrAg -1.20 (0.17 to 18) vs HBsAg -1.97 (0.16 to 18)
- HBV RNA -1.93 (0.14 to 21) vs HBsAg -1.86 (0.15 to 21)
Reductions in concentrations of different markers tended to correlate within individual patients.
Larger phase 2b studies of JNJ-3989 plus NAs are underway in people with chronic HBV infection.
References
1. Gane E, Locarnini S, Lim TH, et al. Short interfering RNA JNJ-3989 combination therapy in chronic hepatitis B shows potent reduction of all viral markers but no correlate was identified for HBsAg reduction and baseline factors. EASL International Liver Congress, June 23-26, 2021. Abstract OS-1430.
2. ClinicalTrials.gov. Study of ARO-HBV in normal adult volunteers and patients with hepatitis B virus (HBV). ClinicalTrials.gov identifier NCT03365947. https://clinicaltrials.gov/ct2/show/NCT03365947
Short interfering RNA JNJ-3989 combination therapy in chronic
hepatitis B shows potent reduction of all viral markers but no correlate was identified for HBsAg reduction and baseline factors


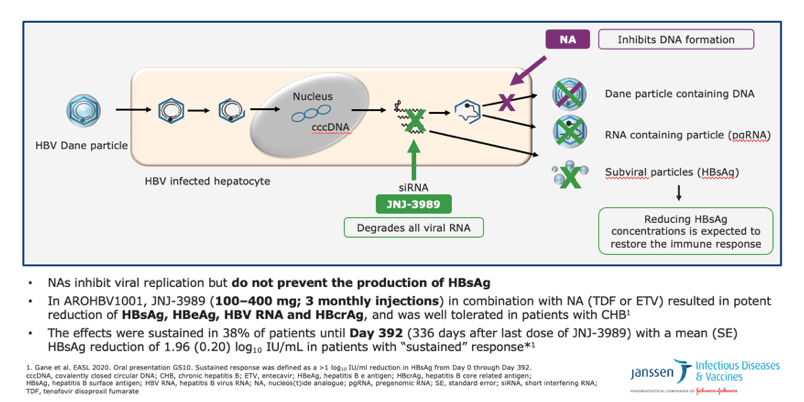
•This diagram summarises the mechanism of action of JNJ-3989 in combination with nucleos(t)ide analogues (NAs)
•siRNA therapy silences HBV RNA transcripts from cccDNA and HBV RNA derived from integrated HBV DNA, thereby inhibiting HBV protein synthesis
•Reducing levels of HBsAg is expected to facilitate restoration of the host immune response
•At EASL 2020, we presented follow-up data at 48 weeks after the last dose of JNJ 3989, demonstrating excellent safety and tolerability and sustained reductions in HBsAg and all other viral parameters from Day 0 through Day 392
•Today, I will report the results of a secondary analysis of the AROHBV1001 study

•Part two of the AROHBV1001 study was an open-label, multiple-ascending dose study to assess the efficacy and safety of JNJ-3989 in patients with chronic hepatitis B
•Patients with chronic hepatitis B, including those who were HBeAg-positive or HBeAg-negative, NA-experienced or NA-naïve, received three subcutaneous doses of JNJ-3989, 100, 200, 300 or 400 mg, administered on Days 0, 28 and 56 were included in the analyses presented here
•All patients received NAs throughout the treatment and follow-up period
•Patients were seen at regular intervals until 48 weeks following the final dose of JNJ 3989
•Serum viral parameters were assessed at each study visit
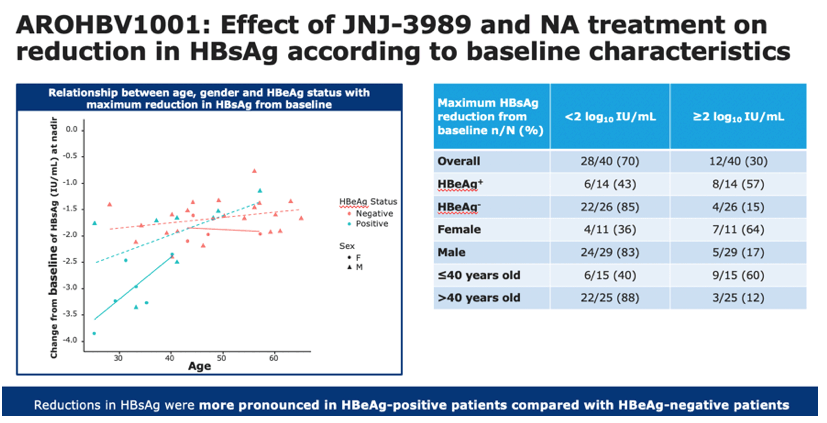
•This figure depicts individual changes at the nadir in HBsAg levels from baseline, according to baseline factors of age, sex and HBeAg status
•Red data points represent HBeAg-negative patients, green data points represent HBeAg-positive patients. Females are represented by circles, and males are represented by triangles
•Reductions in HBsAg were greater in patients who were female, younger than 40 years old and who were HBeAg-positive
In HBeAg-negative patients, there were no apparent associations between HBsAg reductions and age or sex
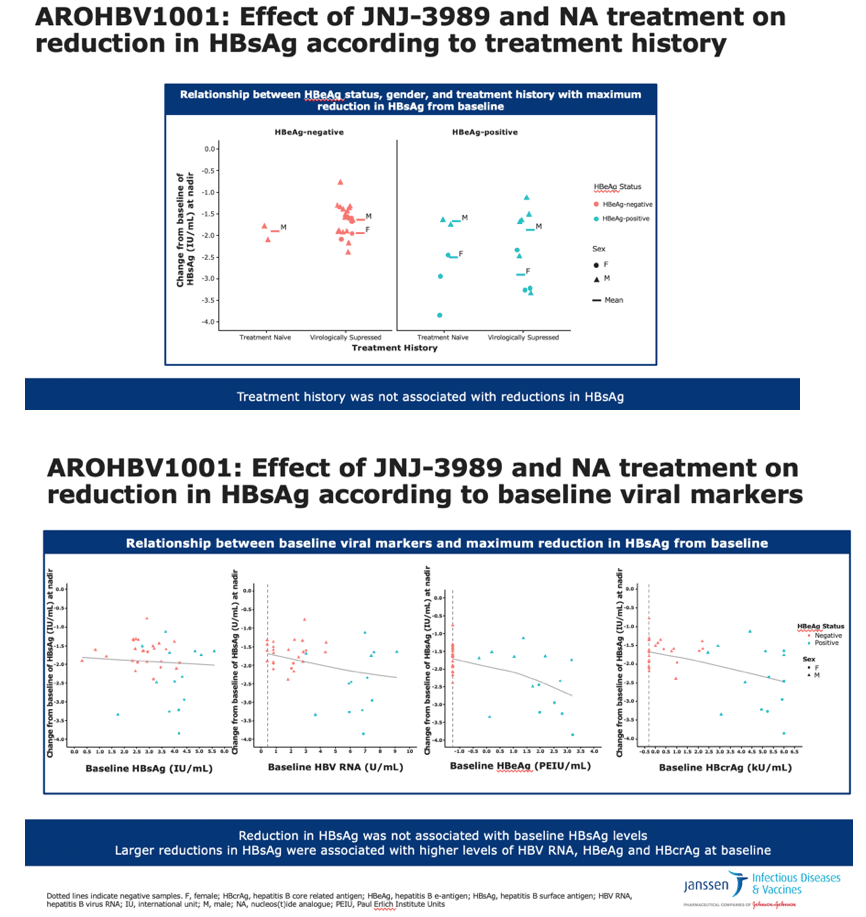
•This slide evaluates the relationship between baseline viral markers and maximum changes in HBsAg levels following treatment with JNJ-3989 and NA
•Reductions in HBsAg levels following JNJ-3989 were not related to baseline levels of HBsAg, but were greater in patients with higher baseline levels of HBV RNA, HBeAg and HBcrAg
•Notably, higher levels of HBcrAg and HBV RNA were correlated to HBeAg-positivity
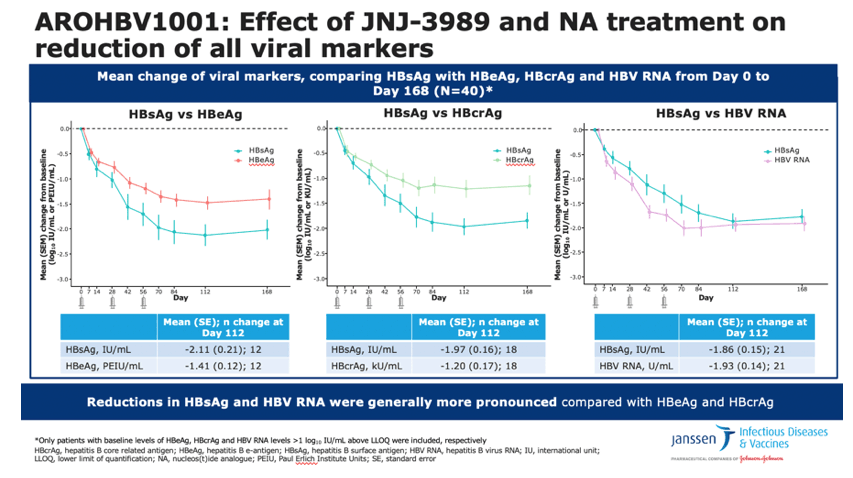
•These figures compare the mean reduction in serum HBsAg levels following JNJ 3989 therapy with mean reductions in HBeAg, HBcrAg and HBV RNA
•Separate analyses include only those patients with detectable levels of that respective marker
•Mean reductions in HBsAg were similar to HBV RNA, but were more pronounced than mean reductions in HBeAg and HBcrAg levels
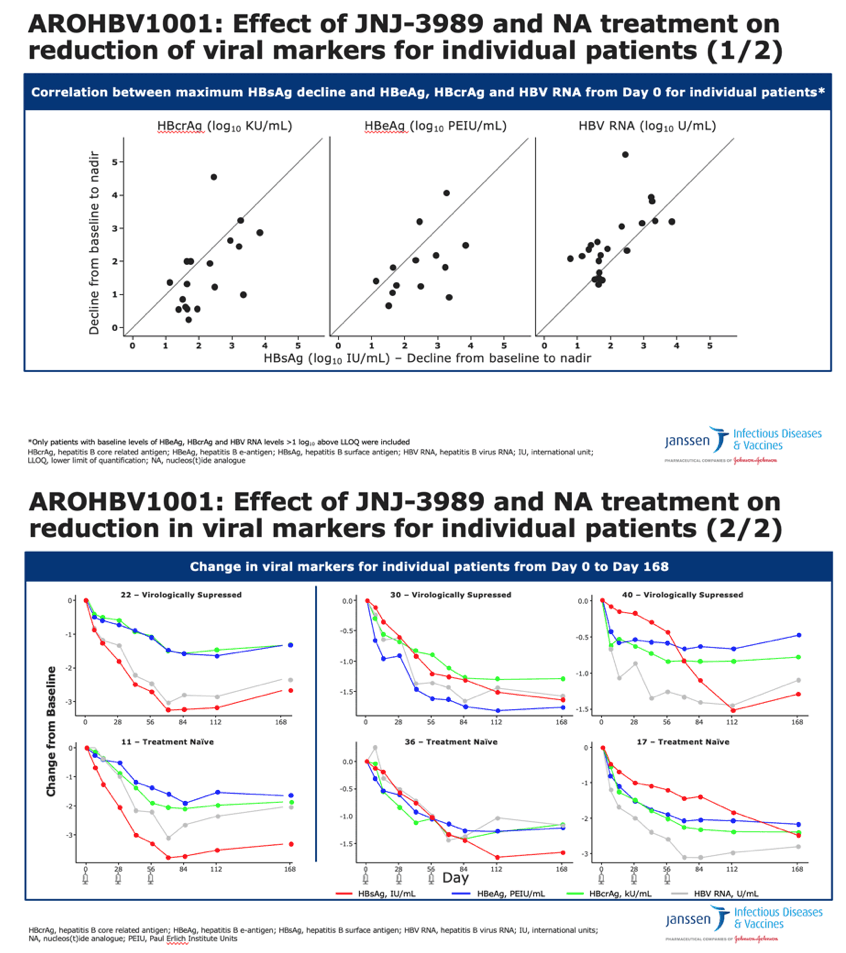
•The point made on the previous slide is further illustrated with individual patient profiles. Here, the two profiles on the left rather reflect the typical pattern, with a more pronounced decline for HBsAg and HBV RNA, while the four panels on the right represent that other patterns were also observed
•It is interesting to note, that reductions of viral markers generally correlated between patients, individual differences were noted

|
| |
|
 |
 |
|
|