| |
ART & Cervical Cancer Women with HIV Risk
|
| |
| |
Download the PDF here
Download the PDF here
Early ART initiation and sustained adherence is likely to reduce incidence and progression of SIL and CIN and ultimately incidence of invasive cervical cancer. Future cohort studies should aim to confirm this possible effect.
The effect of ART on cervical cancer precursor lesions
In the Lancet HIV, Helen Kelly and colleagues1 present a meta-analysis of the effect of antiretroviral therapy (ART) on high-risk human papillomavirus (HPV)-induced cervical lesions in women living with HIV.1 As the authors note, cervical cancer is the most common cancer affecting women in low-income and middle-income countries (LMIC)2 and the most common cancer in women living with HIV.3 For this reason, most HIV management guidelines recommend frequent colposcopy for this population,4 an unpleasant procedure for patients and a resource burden on many health systems.
Cervical cancer is caused by a persistent high-risk HPV infection and develops through a series of well defined precursor lesions, named cervical intraepithelial neoplasia (CIN). CIN lesions are graded 1-3 according to the degree of epithelial atypia. Low-grade CIN (CIN1/2) mainly result from a productive HPV infection and are likely to regress after virus clearance. High-grade CIN lesions (CIN2+) harbour a so-called transforming HPV infection and can develop within 3 years after a persistent high-risk HPV infection. Progression to cervical cancer might take another 10-30 years.5 Infection with HIV is an important risk factor for a persistent high-risk HPV infection and the development of CIN2+ and cervical cancer.6 The high prevalence of high-risk HPV in women living with HIV and the assumed increased cancer risk indicate that a better understanding of the associations between viral infections, treatment, and cervical pathogenesis is needed to allow for effective cancer prevention strategies in this population.
In their meta-analysis, Kelly and colleagues aimed to review and summarise the evidence on the association of ART with high-risk HPV prevalence, and with CIN2+ prevalence, incidence, progression, and regression. Secondly the role of HIV-related cofactors that might modify these associations, such as duration and timing of ART, initiation of ART, immune suppression, and recovery were investigated. The researchers unveiled some findings one might expect on the basis of the ART attributed immune restoration effect. Women living with HIV on ART have a lower prevalence of both high-risk HPV and CIN2+ lesions. Furthermore and as expected, a reduction in the incidence and progression of CIN2+ lesions were seen in women on ART, and an increase in regression of CIN2+ lesions. Notably, these effects remained after adjusting for immune restoration indicators such as CD4 cell count and duration of ART use.
Strong points of this study are the subanalysis on the level of timeperiod and geographical location of the included studies. Some striking discrepancies caused by study heterogeneity were brought to light. On the one hand, studies from Africa, Europe, and North America overall found a preventive effect of ART on cervical lesion incidence and progression and promotion of regression. On the other hand, studies from Latin America and Asia reported an overall increased risk of high-risk HPV and CIN2+ lesions among women on ART compared with treatment-naive women. The latter is counterintuitive if one assumes that ART induced immune restoration would reduce high-risk HPV-associated morbidity. The authors propose the explanation that the Latin American and Asian studies involved a generation of women who might have started ART under older guidelines at lower CD4 cell count thresholds and therefore might not have fully recovered their HPV-specific mucosal immune response. Kelly and colleagues mention that there were fewer studies available from Asia and Latin America and most were cross-sectional in design. Still, these heterogeneous findings again indicate that ART initiation irrespective of CD4 cell count in combination with sustained adherence are important for immune recovery and reduce high-risk HPV-associated morbidity.
Kelly and colleagues1 also offer some practical lessons. Apart from the importance of commencing ART as soon as possible, women with low or unknown nadir CD4 counts should be screened more frequently. Future studies will have to elucidate which women can be screened less frequently on the basis of markers indicating successful immune restoration. Triaging women for colposcopy would relieve overburdened health systems and might reduce unnecessary procedures.
Particularly in LMIC with high prevalence of high-risk HPV and HIV, implementation of effective cytology-based or HPV-based screening programmes has proven difficult. Research shows7, 8 that molecular methylation tests aimed at the detection of lesions resulting from a transforming high-risk HPV infection (high-grade CIN and cancer), might provide an interesting selection method for women living with HIV in LMIC. Further development of rapid point-of-care tests would allow women to be (self) screened and depending on local facilities potentially treated in one day, thereby preventing loss to follow-up.
Furthermore, future research in the line of Kelly and colleagues' could be applied to anal cancer screening in HIV-positive men who have sex with men. Triaging men for high resolution anoscopy to screen for anal precursor lesions would be welcomed for the same reasons.9
-----------------------------
Cervical cancer incidence stratified by age in women living with HIV compared with the general population in the United States, 2002-2016
May 27, 2021
Abstract
Objective:
Recommendations for the age of initiating screening for cervical cancer in women living with HIV (WLHIV) in the United States have not changed since 1995 when all women (regardless of immune status) were screened for cervical cancer from the age of onset of sexual activity, which often occurs in adolescence. By 2009, recognizing the lack of benefit as well as harms in screening young women, guidelines were revised to initiate cervical cancer screening for the general population at age 21. By comparing cervical cancer incidence in young WLHIV to that of the general population, we assessed the potential for increasing the recommended age of initiating cervical cancer screening in WLHIV.
Design:
We compared age-specific invasive cervical cancer (ICC) rates among WLHIV to the general population in the United States HIV/AIDS Cancer Match Study.
Methods:
We estimated standardized incidence ratios as the observed number of cervical cancer cases among WLHIV divided by the expected number, standardized to the general population by age, race/ethnicity, registry and calendar year.
Results:
ICC rates among WLHIV were elevated across all age groups between ages 25-54 (SIR=3.80; 95%CI 3.48, 4.15), but there were zero cases among ages <25.
Introduction
Cervical cancer was designated as an AIDS-defining malignancy in 1993 when it was noted that women living with HIV (WLHIV) were more likely to be diagnosed with cervical cancer at a younger age and more advanced stage compared with the general population.[1] WLHIV continue to have an increased incidence of cervical cancer, as well as other human papillomavirus (HPV)-related malignancies, despite advances in HIV care including immune-reconstitution associated with use of combined antiretroviral therapy (cART).[2]
The United States Public Health Service/Infectious Disease Society of America (USPHS/IDSA) and the American College of Obstetrics and Gynecology (ACOG) guidelines for the age of initiating screening for cervical cancer in WLHIV have not changed since 1995; the guidelines recommend that all WLHIV be screened for cervical cancer from the age of onset of sexual activity, which often occurs in adolescence.[3,4]
Recommendations from ACOG and American Cancer Society for the age to initiate cervical cancer screening in the general population have evolved from initiation of cervical cancer screening at onset of sexual activity or age 18 (1975)[5] to within 3 years of onset of sexual activity or age 21 (2002)[5] to age 21 regardless of age of onset of sexual activity (2009).[6] The 2002 revisions reflect the increasing knowledge of the natural history of HPV from studies conducted in the 1990s, including the high rates of regression of HPV infection and HPV-associated cervical abnormalities in young women.[5] The 2009 revisions were based on 1) observations that cervical cancer rates in adolescents were unchanged from the 1970s through the 2000s[7] and 2) the lack of evidence demonstrating that screening women aged 22-24 reduced the incidence of cervical cancer at ages 25-29.[8] The 2020 recommendations for cervical cancer screening initiation from the Guidelines for the Prevention and Treatment of Opportunistic Infections in Adults and Adolescents with HIV are unchanged from 2009 and state that adolescents with HIV should continue to undergo screening after the onset of sexual activity and not wait until 21 years of age.[9] [10][11]
Current recommendations for age of initiating cervical cancer screening prevention are based on risk of preventable incident cancer and reflect similar management for people with similar cancer risk. Given that the primary driving force behind the change in age for the initiation of cervical cancer screening in the general population to age 21 was the paucity of cancers diagnosed in women under the age of 25, we evaluated the cervical cancer incidence of young WLHIV. If cervical cancer incidence in the general population and WLHIV under age 25 is similar (i.e., rare), that would provide data to support changing the recommended age of initiating cervical cancer screening to age 21 (instead of adolescence) for WLHIV.
Conclusions:
The absence of ICC among WLHIV <25 years supports initiating cervical cancer screening at age 21, rather than adolescence, to prevent cancers in WLHIV at ages with higher risk of ICC.
Results
The study population included 164,084 WLHIV (64.4% Black, 21.8% Hispanic, 12.7% White and 1.1% other race; Table 1). Heterosexual transmission was the most common route of HIV acquisition (44.5%), followed by injection drug use (20.8%). The median year of HIV report was 2004 and the median age at HIV report was 37.8 years. Compared to WLHIV without cervical cancer, WLHIV with cervical cancer had an earlier date of HIV report (median=2001 vs. 2004). A larger proportion of WLHIV with cervical cancer were Hispanic (26.8% vs. 21.8%) and were women who injected drugs (28.8% vs. 20.7%), and a smaller proportion were White (8.2% vs. 12.7%).
During 2002-2016, 552 ICCs occurred in 1.16 million person-years of follow-up among WLHIV (rate=47.7 per 100,000). By age group, the highest incidence rates occurred among 40-44 and 35-39-year-olds (rate=66.1 and 64.5 per 100,000, respectively) (Table 2). Zero cases of invasive cervical cancer occurred among <25-year-old WLHIV during 69,900 person-years of follow-up (SIR=0; 95% CI (0, 7.1)).
When compared to the general population, rates of cervical cancer were elevated significantly 3.4-times overall (95% CI 3.13-3.70). By age, the SIR was the highest among 25-29-year-olds (n=20 cases; SIR=5.34; 95% CI 3.26-8.25) and decreased with increasing age (Figure 1). Cervical cancer rates among WLHIV were significantly elevated across all age groups between ages 25-54 (SIR=3.80; 95% CI 3.48-5.15), as well as among 60-65-year-olds (SIR=2.55; 95% CI 1.56-3.94).
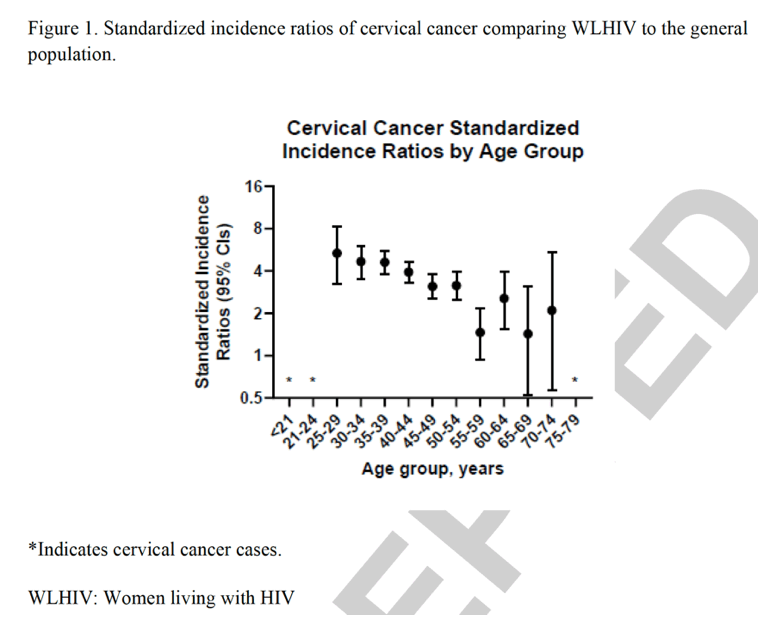
|
|
| |
| |
|
|
|