 |
 |
 |
| |
Switch to DTG/3TC Noninferior to Maintaining TAF Regimen for 144 Weeks
|
| |
| |
IDWeek, September 29-October 3, 2021
Mark Mascolini
Switching to single-tablet dolutegravir/lamivudine (DTG/3TC) proved virologically noninferior to maintaining a 3- or 4-drug regimen including tenofovir alafenamide (TAF) through 144 weeks in the randomized open-label TANGO trial [1]. Overall side effect rates were minimally higher with DTG/3TC, and over 10% in both groups gained more than 10% in weight.
DTG/3TC was an effective first-line regimen through 144 weeks in GEMINI-1 and -2 and a reliable switch option for 48 weeks in people taking an effective regimen with no history of virologic failure in the SALSA study.
TANGO is an ongoing trial of maintaining a 3- or 4-drug TAF-containing regimen or substituting DTG/3TC for the TAF combo. Participants in this international trial are adults who had a viral load below 50 copies for at least 6 months on a TAF regimen. They had no history of virologic failure and no resistance to nucleoside/nucleotide analogs or an integrase inhibitor.
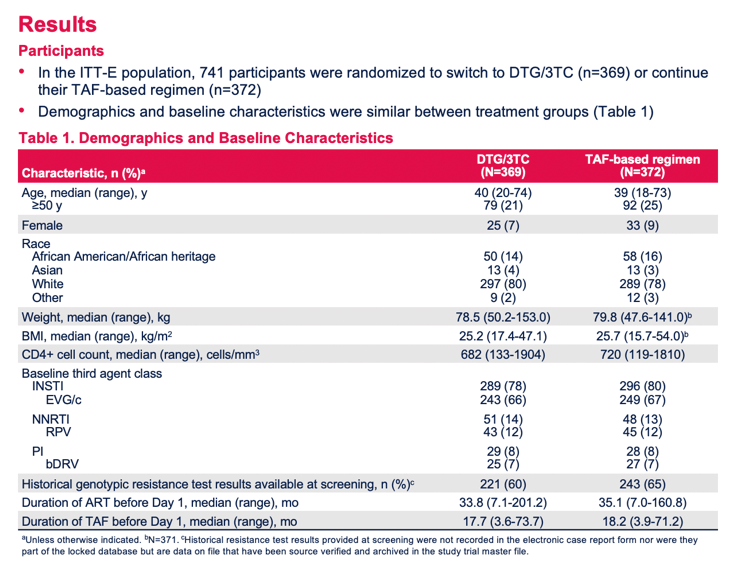
Researchers randomized 369 people to switch to DTG/3TC and 372 to keep their TAF combination. At week 144 everyone in the TAF group could change to DTG/3TC. When TANGO began, the DTG/3TC and TAF groups had a similar young median age (40 and 39 years, 21% and 25% aged 50 years or older), similar low proportions of women (7% and 9%), similar high proportions of whites (80% and 78%), and low proportions of blacks (14% and 16%). Median CD4 count lay above 680 in both groups. Two thirds in each group were taking an elvitegravir/cobicistat TAF regimen, and 12% in each group were taking the nonnucleoside rilpivirine.
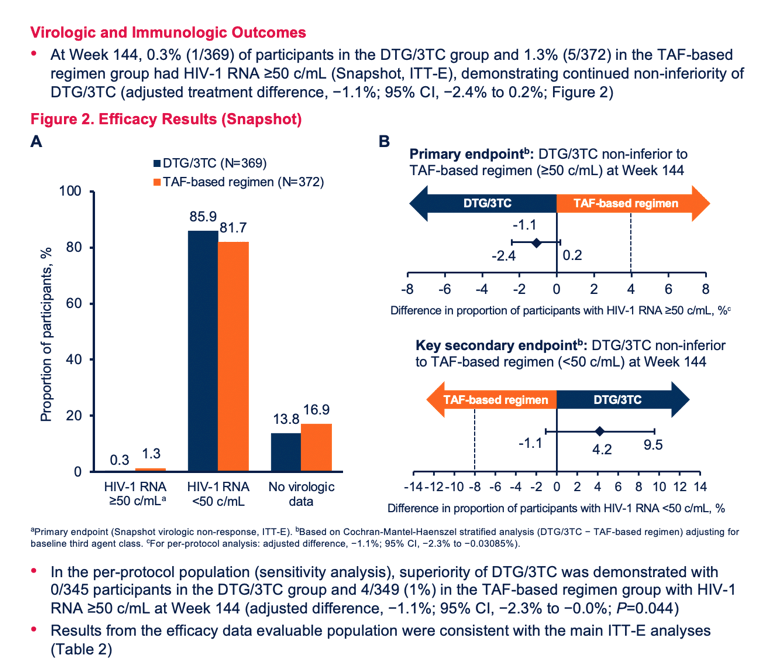
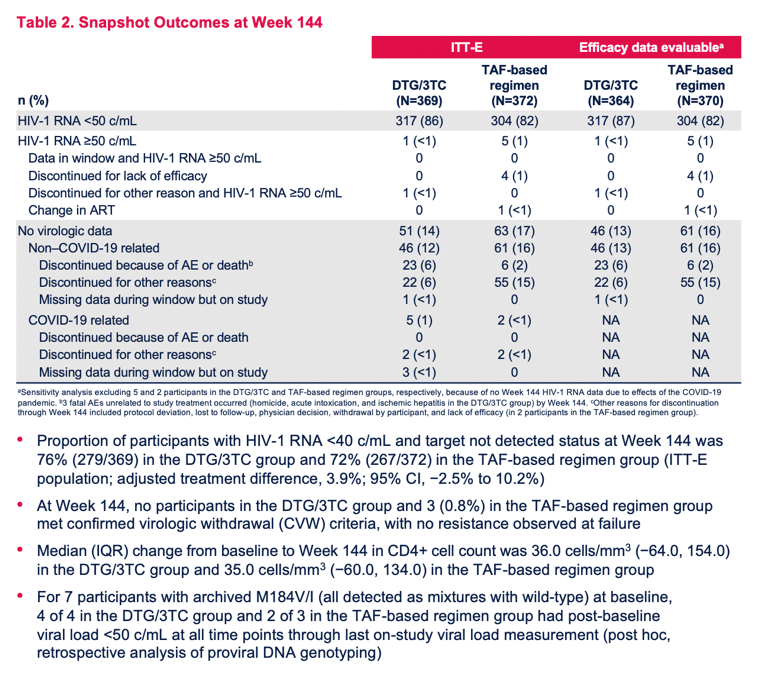
In a week-144 intention-to-treat-exposed analysis (including everyone who took at least one dose after randomization), 0.3% (1 of 369 people) in the DTG/3TC group and 1.3% (5 of 372) in the TAF-maintenance group had a viral load above 50 copies, a result confirming virologic noninferiority of switching to DTG/3TC compared with continuing an effective TAF regimen. While 85.9% of people randomized to DTG/3TC had a 144-week viral load below 50 copies, 81.7% in the TAF arm met that standard.
No one assigned to the DTG/3TC group met criteria for confirmed virologic withdrawal, compared with 3 people (0.8%) in the TAF group. Resistance analysis at failure in the TAF arm disclosed no resistance mutations. Among 7 people who had evidence of the M184V/I mutation conferring resistance to 3TC before randomization, 4 of 4 in the DTG/3TC group and 2 of 3 in the TAF group had a viral load below 50 copies at every measure through the last viral load assessed for this analysis.
Median CD4 gain in the two study groups virtually overlapped: 36.0 (interquartile range [IQR] -64 to 154) with DTG/3TC and 35.0 (IQR -60 to 134) with continued TAF.
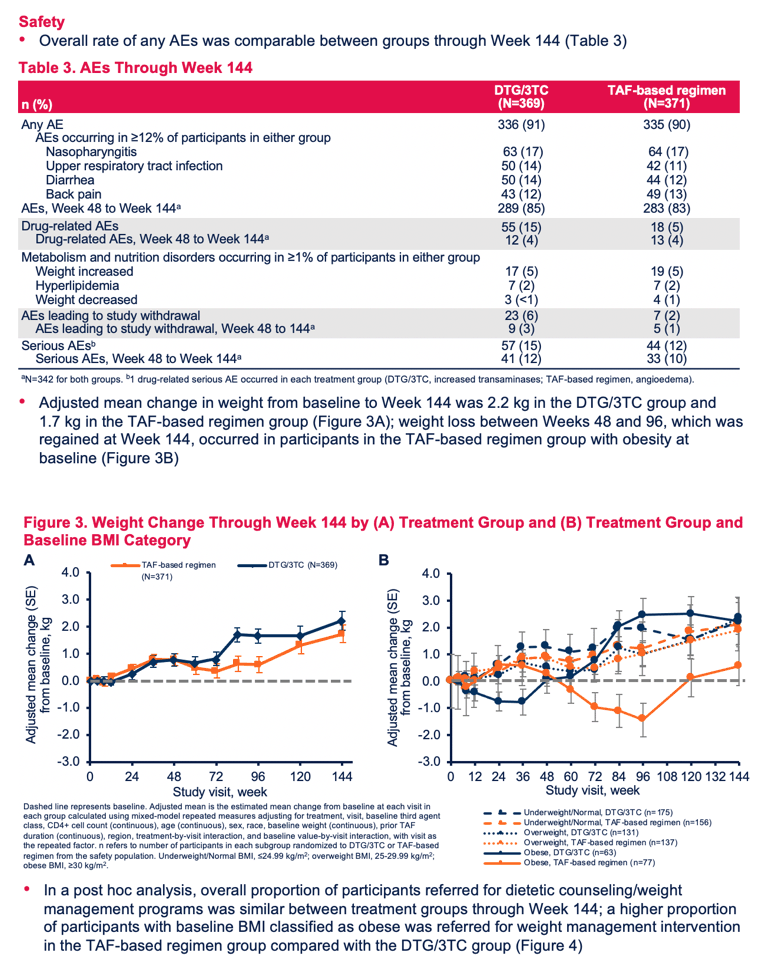
Through 144 weeks, 15% in the DTG/3TC group and 5% in the TAF group had a drug-related adverse event. In both groups 4% had a drug-related adverse event between week 48 and week 144. Adverse events leading to study withdrawal numbered 23 (6%) in the DTG/3TC arm and 7 (2%) in the TAF arm. Numbers and percentages of adverse events leading to withdrawal between week 48 and week 144 were 9 (3%) with DTG/3TC and 5 (1%) with continued TAF. Serious adverse event rates through week 144 were 15% with DTG/3TC and 12% with TAF.
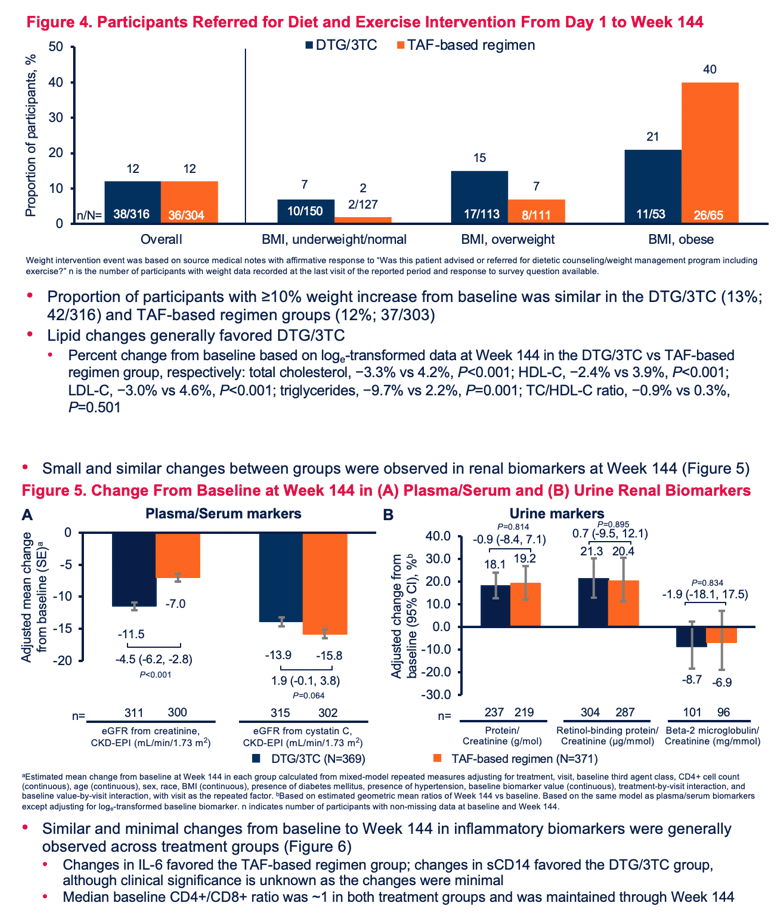
Adjusted average weight gain through 144 weeks measured 2.2 kg (4.85 lb) with DTG/3TC and 1.7 kg (3.75 lb) with TAF. Proportions of participants with 10% or more weight gain through 144 weeks were 13% with DTG/3TC and 12% with TAF. Total-to-HDL cholesterol ratio fell (improved) in both groups in 144 weeks, by 0.9% with DTG/3TC and by 0.3% with TAF, a nonsignificant difference (P = 0.501).
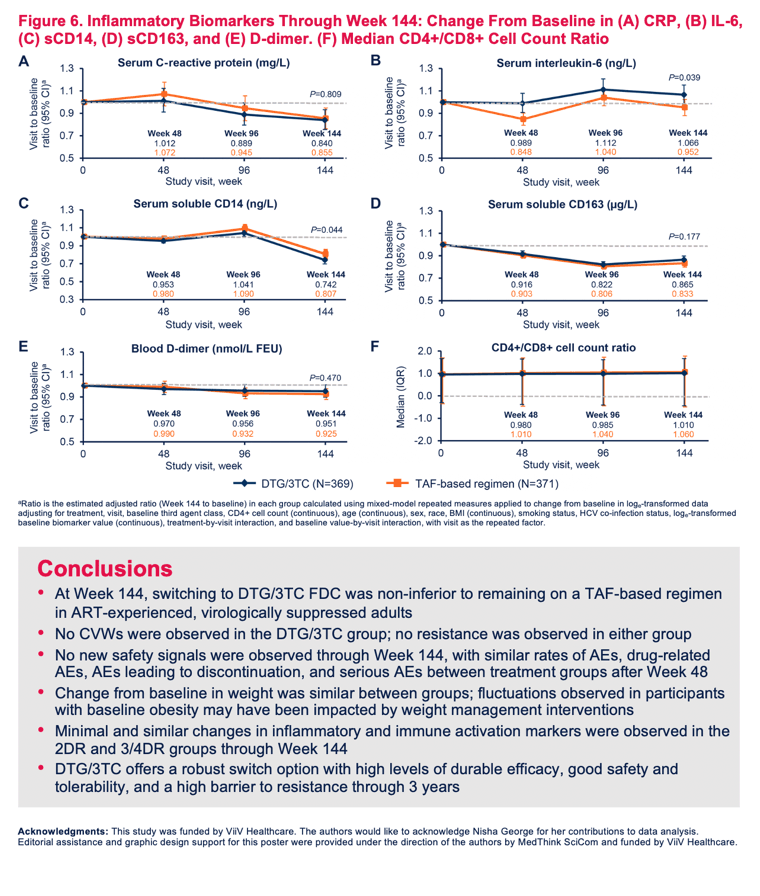
Levels of four inflammatory markers dwindled marginally in both study arms, with no significant difference between groups through week 144: C- reactive protein, soluble CD14, soluble CD163, and D-dimer. Interleukin 6 rose modestly with DTG/3TC and stayed about the same with continued TAF (visit-to-baseline ratio 1.066 vs 0.952, P = 0.039).
Reference
1. Osiyemi O, Ajana F, Bisshop F, et al. Switching to DTG/3TC fixed-dose combination (FDC) is non-inferior to continuing a TAF-based regimen in maintaining virologic suppression through 144 weeks (TANGO Study). IDWeek, September 29-October 3, 2021. Abstract 900.

|
| |
|
 |
 |
|
|