 |
 |
 |
| |
Summary from virtual CROI 2022 for HIV and liver disease
Hepatitis elimination for 2030 on track?
Is fatty liver disease a growing concern?
New insights from CROI 2022.
|
| |
| |
Jurgen K. Rockstroh M.D., Professor of Medicine
University of Bonn, Germany
Correspondence:
Prof. Dr. Jurgen K. Rockstroh
Department of Medicine I
University Hospital Bonn
Venusberg-Campus 1
53127 Bonn
Germany
Introduction
Unfortunately, 2022 has now become the third year in row where CROI had to take place as a virtual conference meeting. This however, has not strongly impacted interest or participation in the CROI meeting but has been accompanied by a significantly lower number of abstract submissions in the liver disease field. As a consequence of shifted interests but also rechanneled research money and working capacity, new results around COVID-19 vaccines and antivirals have dominated the scientific submissions to CROI with many subsequent oral abstract sessions covering various new aspects of COVID-19 research. The oral abstract session for hepatitis research was integrated into the session on tuberculosis, opportunistic infections, and hepatitis. Indeed, there was only one oral abstract on translational HBV research, two on HCV, one on talaromycosis and five on various aspects around tuberculosis in that particular session. Nevertheless, there were still numerous interesting abstract submissions which could be found in the poster sessions focusing on various aspects of liver disease in HIV. Abstracts are available within the abstract book on the CROI website under the resource button (see: https://www.croiconference.org/wp-content/uploads/sites/2/resources/2022/croi2022-abstract-ebook.pdf). The main topic in the oral abstract session was clearly HCV elimination targets in light of the COVID-19 pandemic but also availability of easy to administer all oral HCV DAA-based therapies (1,2). Whereas, the US appears to struggle even more under the impact of COVID-19 on hepatitis services to stay on target for reaching the HCV elimination goals, Western Europe and Australia are well on track in their HIV/HCV coinfected population to reach HCV elimination by 2030. Another important HCV presentation highlighted the advantages of universal HCV screening among pregnant people attending care in Western Pennsylvania versus risk-based HCV screening (3). Other important topics discussed at this year CROI included results of a randomized HBV vaccination trial, new HBV infections in an HIV cohort in Texas, new long-acting tenofovir formulation for HBV therapy, HCV elimination in MSM, DAA salvage therapy, clinical outcomes after SVR and various aspects around fatty liver disease (4-14). The following summary aims at highlighting some of the exciting new data reported at CROI 2022 and to initiate a discussion about their usefulness and relevance for clinical management of liver disease in HIV-coinfected individuals.
Hepatitis B
Implementation of universal neonatal vaccination program against hepatitis B virus (HBV) has significantly reduced HBV seroprevalence in the general population as well as in people living with HIV (PLWH). PLWH are well-known to show lower response rates to HBV vaccination particularly in the setting of ongoing HIV-replication or low CD4-counts. At this year CROI a randomized controlled trial was presented from Taiwan which investigated the serological responses to three standard- (20-μg) or double-dose (40-μg) HBV revaccination among HIV-positive and HIV-negative men who have sex with men (MSM) (4). Only PLWH on stable antiretroviral therapy were included. Participants were randomized in a 1:1 ratio (stratified by CD4 count for PLWH) to receive three standard or double doses of HBV vaccine that were administered at Weeks 0, 4, and 24. 249 (71%) HIV-positive MSM and 104 (29%) HIV-negative MSM with a mean age of 27.8 years were enrolled with 178 and 175 in two arms, respectively. The two groups were well balanced in terms of clinical characteristics. In PLWH, 70% had CD4 counts > 500 cells/mm3 and 95% were virally suppressed (HIV RNA load <50 copies/ml). The serological response rates are shown in figure 1a and b.
Figure 1a and b: Proportion of Serological Response (anti-HBs ≥10 mIU/ml) and proportion of high-titer response (anti-HBs ≥100 mIU/ml)
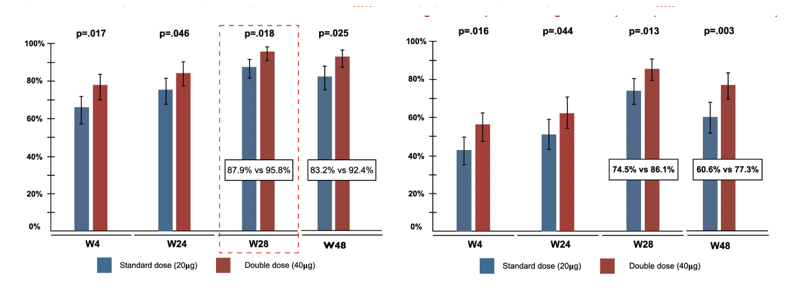
In multivariate logistic regression analysis, double-dose HBV revaccination and baseline anti-HBs titer were significantly associated with serological response at Week 28. For high-titer responses, double-dose HBV revaccination, younger age, and baseline anti-HBs titer were significantly associated with serological response at Week 48.
In conclusion, this study demonstrates that revaccination with three double doses of HBV vaccine achieves higher serological responses than with three standard-doses of HBV vaccine among MSM who were born in the era of universal neonatal HBV vaccination. Virologically suppressed PLWH with CD4-counts >500 cells/mm3 had similarly responses after revaccination as healthy MSM. This once again underlines that HBV vaccination in newly diagnosed PLWH is done at best when viral suppression has occurred after starting ART.
Despite all data highlighting the need for HBV vaccination in PLWH, at each CROI data are being presented which document low HBV vaccination rates in real-life settings. Most alarmingly, only 9% (583/ 6506) of PLWH from two Texas HIV centers without documented HBsAb+ received 3 doses of hepatitis B vaccine in 12-month period underlining the need to better implement HBV vaccination as recommended by all national and international guidelines (15). Moreover, only half of those who received 3-dose vaccine developed immunity among those who were tested, indicating that there is a need for developing better vaccines and vaccination strategies. Indeed, double dose vaccination as described above may be one way to improve vaccination response rates.
Vaccines and hepatitis B virus (HBV) specific antiretrovirals (ART) may offer protection for people living with HIV (PLWH) against acquiring new HBV infection. At CROI 2022 a study was presented which examined the incidence of HBV and factors which predicted risk for acquiring infection among PLWH in the US (5). The study included all PLWH with a minimum of 1 up to 9 years of follow-up from 1/1/11 to 12/31/18. Patients were excluded if no HIV viral load or CD4 count were available as well as all patients with signs of chronic or occult HBV infection. Out of 26,152 PLWH, 12,285 were included (72.3% male, 56.9% Black), 35.2% had CD4 <200 cells/µL, 12.3% HIV viral load <50 copies/mL, 42.6% were HBV immune at study entry. Within 2 years of study entry, 80.6% were on an HBV-specific HIV regimens. Overall, 0.49% (n= 60, 70% male, 77% Black; 65% non-immune and 52% with CD4 <200 cell/µL at baseline) developed incident hepatitis B during follow-up. PLWH who are unvaccinated for hepatitis B clearly remain at higher risk for new HBV infections as shown in figure 2.
Figure 2: Incidence of new hepatitis B infection based on HBsAg+ stratified by baseline HBsAb+
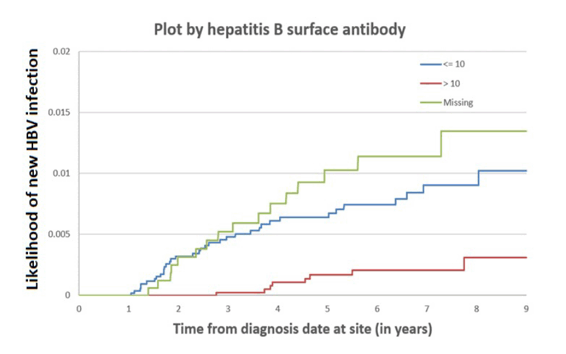
Of note in contrast to other studies the authors did not observe a protective effect of HBV specific medication at baseline for development of new HBV infection in follow-up. The poster however, did not provide information which HBV specific medication was used. Prior studies from the Netherlands demonstrated a persistent risk for new HBV infections in PLWH receiving 3TC as sole HBV active agent but not necessarily in tenofovir treated patients (16). Moreover, the US investigators showed that patients with HCV infection were at increased risk for acquiring HBV infection underlining the need for HBV vaccination in this population subgroup (figure 3).
Figure 3: Incidence of New HBV Infection based on +HBsAg stratified by HCV
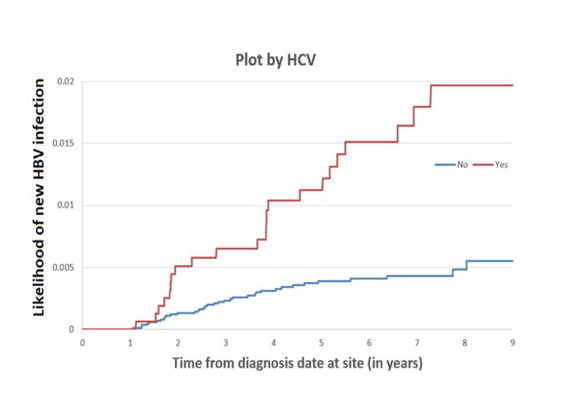
In conclusion, this study underlines the importance of recommending HBV vaccination in HBV seronegative PLWH even if they are on dual active HIV/HBV antiviral therapy.
Overall, hardly any data was presented on new HBV treatment agents. Of note, however was an animal study which reported on a nanoformulated tenofovir (TFV) prodrug which was able to suppress HBV DNA in humanized and transgenic mice for three months following a single intramuscular injection (6). For this study a lipophilic TFV prodrug, M1TFV, was synthesized and nanoformulated into stable poloxamer 407 stabilized aqueous nanocrystals (NM1TFV) by high pressure homogenization. Solid drug nanocrystals of TAF (NTAF) were produced and used as controls. Formulation efficacy was evaluated in two mouse models (HBV-infected humanized liver TK-NOG mice and HBV transgenic Tg05 mice) following a single intramuscular injection of 168 mg/kg TFV equivalents of either NM1TFV or NTAF. HBV DNA levels in peripheral blood were assessed biweekly for 12 weeks. HBV markers HBcAg and HBsAg were evaluated on stained liver sections of TK-NOG mice. Drug levels were quantified by mass spectrometry. A single intramuscular injection of NM1TFV to HBV infected humanized TK-NOG mice suppressed viral DNA in peripheral blood and liver for three months. Levels of HBcAg and HBsAg were equally suppressed over three months in NM1TFV treated mice. Overall, NM1TFV formulations were well tolerated and no evidence of muscle degeneration, basophil or eosinophil infiltration, cellulitis or abscesses were observed at the injection site. The authors conclude that NM1TFV can be developed as a therapy for chronic HBV infection. Just as in other medical areas development of long-acting treatment options could be of considerable interest in HBV patients who require potentially lifelong treatment of chronic HBV infection and are facing adherence challenges.
Hepatitis C
In 2016, WHO proposed eliminating hepatitis C as a public health threat by 2030, targeting an 80% reduction in new chronic infections and a 65% reduction in mortality from 2015 levels. Most countries will miss these targets. At this year CROI modeling data on the impact of the COVID-19 pandemic on HCV elimination in the US was presented (1). The researcher team applied their model to three scenarios: Scenario 1: Status quo—No disruption to HCV treatment uptake, Scenario 2: One-year COVID-19 disruption—30% reduction in HCV treatment uptake from 2020 to 2021 (the rate observed in national data) and Scenario 3: Two-year COVID-19 disruption—30% reduction in treatment uptake from 2020 to 2022. Overall, there were 67,267 (95%CI 23,433-114,471) estimated new HCV infections in 2015. Under all scenarios the 80% incidence reduction target was missed (29.7% reduction by 2030 even with no disruption to treatment. Modeling resulted in 990 (95% 417-1,330) additional new infections from 2015-2030 for the 1-year disruption, and 1,933 (95% 800-2,599) additional new infections for the 2-year disruption. Similarly, all projections failed to meet the mortality target by 2030. Indeed, the model projected a 20.6% decline of HCV-related mortality from 2015 to 2030 in the 2-year disruption scenario (95% CI 14.4 to 29.5) which is far away from the proposed WHO target of 65%. By 2030 the model foresaw 847 excess HCV-related deaths in the 1-year disruption scenario (95% CI 645 to 1061) and 1583 excess deaths in the 2-year disruption scenario (95% CI 1210 to 1981). The estimated mortality results are shown in figure 4.
Figure 4: Results: Excess HCV-related deaths by 2030 compared to no COVID-19 disruption.
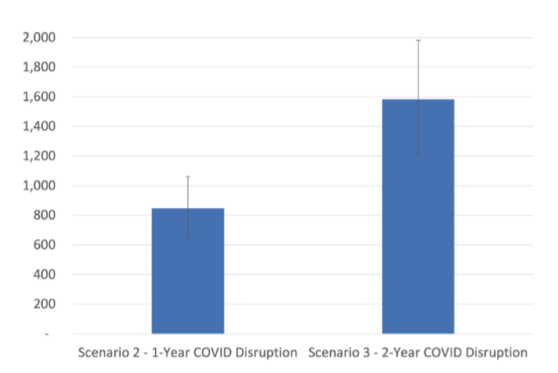
In conclusion, the U.S. is currently not on track to achieve the WHO elimination targets pre-pandemic and has fallen further behind during the COVID-19 pandemic. Increased efforts including scale-up of HCV diagnosis, treatment, and prevention are needed to overcome this unfavorable development.
These results from the US were contrasted by another HCV elimination presentation in the oral hepatitis abstract session from Australia and western Europe (2). Indeed, universal access to direct-acting antivirals (DAA) could help to reduce HCV incidence through a treatment as prevention effect. In this international study, pooled data from 6 cohorts from the International Collaboration on Hepatitis C Elimination in HIV-coinfection (InCHEHC), including data from the Netherlands, Switzerland, Australia, Spain, and France was analyzed between (2010-2019. Within this large cohort effort, the progress towards HCV elimination was monitored, including changes in primary HCV incidence by calendar year and following DAA introduction among people living with HIV (PLHIV). Of 86,250 participants, 45,933 had at least one HCV antibody negative result and a subsequent test. During 248,186 person-years (py), 2,051 incident HCV infections were observed. Incidence decreased from 0.91 per 100 py (95%CI=0.80,1.03) in 2015 to 0.46 per 100 py (95%CI=0.35,0.60) in 2019, reflecting a 50% decrease (see figure 5).
Figure 5: Results: Change in primary HCV incidence after DAA introduction in Europe and Australia.
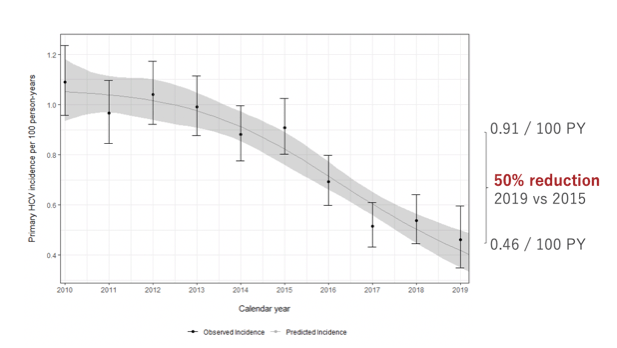
Most impressively, incidence was 0.5/100 PY versus the newly defined target of 5/100,000 PY. The second aim of the study was to assess the effect of DAA on HCV incidence. For this analysis the authors defined three periods of DAA access: No access, restricted access and broad access. Changes in DAA access occurred at different times per country. Piecewise exponential model was applied which included a random effect for country. The drastic drop in HCV incidence was only possible in the setting of broad DAA access (no liver fibrosis level restrictions for receiving DAA therapy) as shown in figure 6.
Figure 6: Predicted HCV incidence per DAA period
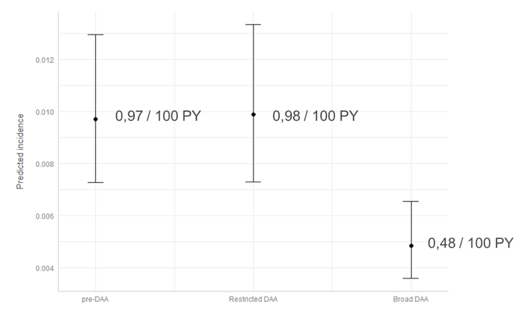
These data suggest that overall, in the pooled analysis countries are on track to meet the WHO elimination incidence target for PLHIV in care by 2030.
Men who have sex with men (MSM) have been identified as one subgroup with continuous hepatitis C virus (HCV) transmission risk due to persistent sexual risk behavior and recreational drug use. On the other side they have also been declared as a target population for HCV micro-elimination efforts as they are well linked to care because of enrolment in PrEP therapy or in case of underlying HIV-infection because of their lifelong HIV treatment. At this year CROI data from the ongoing national German NOCO cohort was presented which collects data on newly acquired HCV infections in MSM since 2014 following the introduction of direct acting antivirals (DAAs) (7). The NOCO cohort consists of patients from six German HIV and hepatitis treatment sites providing care for more than 8000 HIV-positive MSM, and serving as primary care providers and HIV pre-exposure prophylaxis (PrEP) sites. Objective of the current analysis was to study the impact of the COVID-19 pandemic on recently acquired HCV infections and changes in service availability and/or behavior change. Between January 2014, and October 2021, 237 MSM with recently acquired HCV infection were included. The majority were Caucasian (95%), and mean age was 45.3 years (SD 9.57). At HCV diagnosis, median ALT level was 224 U/L (interquartile range, IQR, 86 - 521), and median HCV viral load was 475,000 IU/mL (IQR 66,955 - 3,005,882). The most prevalent HCV genotype were 1a (58.7%), and 4d (16%), respectively. A subgroup of 21 (8.9%) MSM were not co-infected with HIV, of whom 15 (71.4%) were using PrEP. Anti-HCV treatment with DAAs was documented in 165 patients (71.7%), 18 (7.8%) had a spontaneous clearance, and in 47 patients (20.4%) treatment was not started. DAAs were initiated a median 6.6 months (IQR 4 to 9.3) after diagnosis; all treated patients achieved a sustained virologic response (SVR), or treatment was still ongoing (16%). Between 2014-2019 27-36 patients were diagnosed with recently acquired HCV annually. In relation to all HIV-positive MSM under care, the incidence was 0.33 - 0.39% per year with no significant change over time. In 2020, a decline in HCV incidence to 0.28% was observed. In 2021 HCV incidence dropped to 0.02%. In the same period, the number of patients seen in the centers remained stable, and routine HCV testing returned to pre-pandemic levels by the end of 2020. Figure 7 depicts the incidence of HCV in people living with HIV (PLWHI) over the study period.
Figure 7: Incidence of HCV infection in PLWHI in the six Noco Centers. The green bar represents HCV elimination (80% incidence reduction from 2015 baseline).
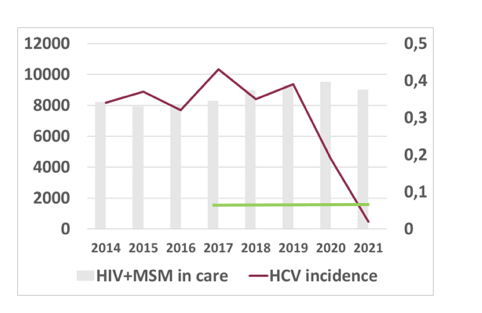
In conclusion, the German NoCo cohort demonstrates stable HCV incidence rates despite a broad use of DAAs up to 2019. In 2021, however, micro-elimination goals were met due to a drop in HCV incidence, possibly due to behavior changes related to the SARS-CoV-2 pandemic and associated containment measures or because of a declining HCV reservoir over time with many MSM successfully treated with DAAs. Interestingly, this is well in line with recent data published at the EACS conference in October 2021 from the Swiss Cohort study which also demonstrated a further reduction in new incident HCV cases among HIV-coinfected MSM (17).
Further interesting HCV elimination data were presented at CROI from Taiwan (8). Taiwan has committed to achieving HCV elimination by 2025 so even earlier than proposed by WHO. HCV testing and antiviral treatments are reimbursed by the National Health Insurance which is the crucial prerequisite for achieving these ambitious elimination goals. The objective of the study presented at CROI was to examine the progress toward HCV microelimination among people living with HIV (PLWH) who had HCV viremia. PLWH seeking care at the National University Hospital Taiwan between 2011 and 2021 received HCV serological testing at least once annually. Those who tested HCV-seropositive at baseline or who received antiviral treatments with achievement of spontaneous clearance or sustained virological response (SVR) underwent HCV RNA testing at least once annually. Between 2019 and 2021, those with episodes of sexually transmitted infections, having achieved spontaneous clearance or (SVR), or elevated aminotransferases underwent HCV RNA testing every 12 weeks for 48 weeks. Annual incidence and prevalence of HCV viremia and HCV seroconversion from 2011 to 2021 are recorded and shown in figure 8.
Figure 8: Trends of prevalence of HCV viremia prevalence in PLWH from Taiwan from 2011 to 2021
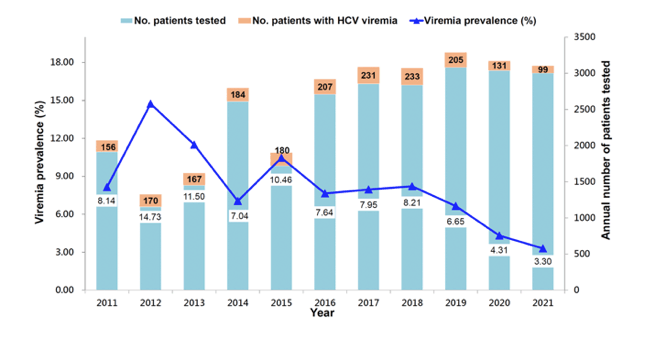
During the 11-year study period, the incidence rate of HCV seroconversion among those testing HCV-seronegative declined from the peak of 25.79 per 1000 person-years of follow-up (PYFU) in 2018 to 8.27 per 1000 PYFU in 2021 (a decrease by 67.9%. The incidence rate of HCV viremia decreased from a peak of 131.6 per 1000 PYFU in 2011 to 20.3 per 1000 PYFU in 2021 (a decrease by 84.6%). The prevalence of HCV viremia declined from 14.73% of PLWH tested in 2012 to 7.04% in 2014 (interferon/ribavirin era), which further declined from 8.21% in 2018 to 3.30% in 2021 (DAA era) (a decrease by 77.6%).
In conclusion, with regular HCV testing and availability of simple to take and better tolerated all oral HCV therapies in Taiwan, the incidence rate and prevalence of HCV viremia declined by 80% among people living with HIV between 2011 and 2021. These results are impressive and clearly underline that in well targeted patient populations with good linkage to care HCV microelimination appears feasible.
Recommendations for HCV screening during pregnancy for a long time have been different between various set of guidelines. More recently, the American College of Obstetrics and Gynecology joined the CDC in recommending universal hepatitis C virus (HCV) screening of pregnant people. Universal HCV screening has been hypothesized to increase the detection of HCV among pregnant people, resulting in fewer undetected perinatal HCV transmissions. At CROI 2022 an interesting study was presented which compared screening and case detection of HCV during risk-based versus universal HCV screening among pregnant people attending care in Western Pennsylvania (3). Universal HCV screening was implemented in June 2020 in Western Pennsylvania using a Best Practice Alert in the electronic medical records (EMR) which provided a reminder if HCV testing was not ordered with the new OB panel and reflex testing for HCV RNA if IgG positive. HCV detection was compared using Fisher's exact test for all pregnant people presenting for prenatal care within one health system during two 12-month time periods: 1) risk-based HCV screening for 12,142 pregnant people (January 1-December 31, 2019) and 2) universal HCV screening for 12,588 pregnant people (July 1, 2020-June 30, 2021). The EMRs were reviewed for HCV testing nine months before and after initiation of prenatal care. Data from January to June 2020 was excluded due to concerns about poor health care utilization and disruptions in operations due to COVID-19. Universal HCV screening resulted in an increase in HCV IgG screening from 23% to 81% of people entering obstetrical care (p<0.001) (see table 1).
Table 1: Results: Risk based versus universal HCV screening among pregnant people
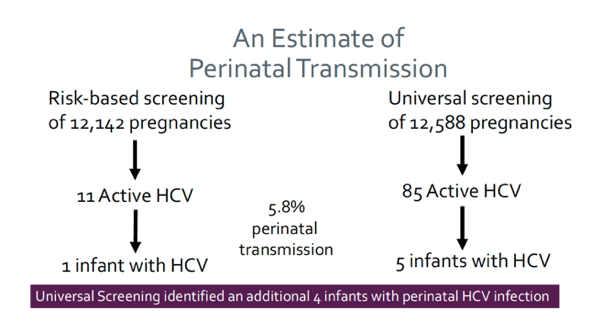
The positivity rate of HCV IgG was higher among those who were tested using risk-based vs. universal screening (5.4% vs. 2.3%) (p<0.001). However, the prevalence of HCV IgG+ people was lower in the overall population using risk-based vs. universal screening (1.2% vs 1.9%) (p<0.001). Utilization of reflex HCV testing resulted in an increase in active HCV detected from 11 (0.091%) to 85 (0.68%) of the obstetrical population during the two 12-month monitoring periods (p<0.001). Given the HCV perinatal transmission rate of 5.8%, an additional 4 infants with perinatal HCV in 12 months were identified with universal screening that had previously gone undetected with risk-based screening within our system. In conclusion, risk-based HCV screening is insensitive for HCV detection within the general obstetric population. The study results strongly support universal HCV screening of pregnant people not only to ensure that the pregnant person is linked to treatment, but also to ensure detection of all cases of perinatal HCV transmission.
Although, HCV cascade of care, HCV elimination and screening for HCV were the main topics at CROI for HCV this year there were also some interesting presentations on treatment issues. One interesting study from Spain looked at efficacy and safety of HCV retreatment with DAA triple therapy consisting of Sofosbuvir/Velpatasvir/Voxilaprevir in HIV/HCV coinfected individuals who previously failed DAA therapy (9). So far data on this combination in HIV/HCV coinfection have been scarce. Patient data was derived from the RUA-HCV registry which is a prospective and mandatory registry of adults (≥18 years) undergoing therapy with DAAs for HCV infection in the region of Madrid since Nov 2014. All data were exported to the Madrid Coinfection Registry (Madrid-Core), created to determine the effectiveness and safety of all-oral DAAs in PLHIV who are HCV coinfected. 56 patients included in Madrid-Core, who were ≥18 years old, re-treated with SOF/VEL/VOX and were scheduled to finish treatment before Jan 1,2021, were included into the analysis. Treatment schedule was SOF/VEL/VOX 1 qd for 12 weeks. Sustained virologic response (SVR) was assessed at 12 weeks after the end of planned treatment. The baseline characteristics of the retreated patients are shown in table 2:
Table 2: Baseline characteristics

The majority of patients had low fibrosis stages. The number of previous DAAs regimens was one in 76.5%, 2 in 15.5%, and three or more in 7.8%. The type of previous regimens included were sofosbuvir/ledipasvir in 50% patients, ombitasvir/paritaprevir/ritonavir plus dasabuvir in 9.4%, sofosbuvir plus daclatasvir in 9.4%, elbasvir/grazoprevir in 9.4%, glecaprevir/pibrentasvir in 9.4% and 13.4% other regimens. SVR rates were 80.4% (95% CI, 68.2%-89.7%) by ITT and 95.7% (95% CI, 85.7%-98.8%) by PP analysis. A total of 9 patients were not included in the PP analysis (2 discontinuations and 7 without data). Liver cirrhosis and genotype did not influence treatment response (SVR by ITT 90% for cirrhosis and 85.8% for G3) (see figure 9).
Figure 9: SVR rate depending on presence of cirrhosis and baseline HCV genotype.
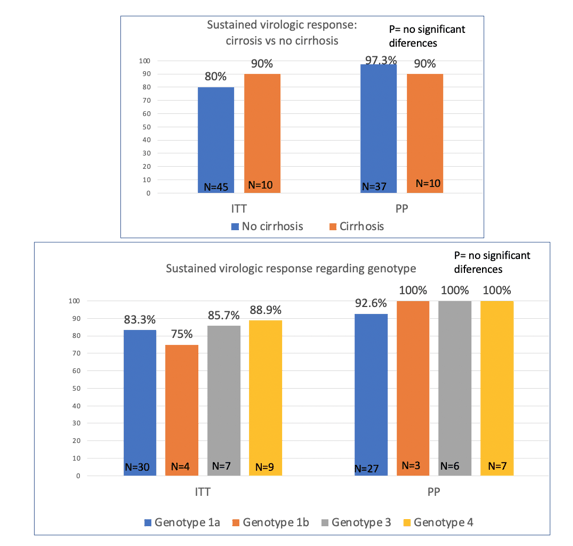
In conclusion, these data clearly demonstrate that SOF/VEL/VOX is a highly effective regimen for treatment of coinfected patients previously failing DAA regimens, across all genotypes and even in the presence of cirrhosis.
Affordable direct-acting antivirals are urgently needed to treat hepatitis C virus (HCV) infection in low and middle-income countries. In order to have affordable DAAs, the Drugs for Neglected Diseases initiative (DNDi) set a goal in 2016 to develop a safe and effective direct-acting antiviral that would be made accessible and affordable to all. To meet this goal, DNDi pursued an alternative drug research and development pathway: one with affordability at its core and founded on collaborations with low-income and middle-income countries (LMICs). In 2016, DNDi established an innovative partnership between the Ministry of Health in Malaysia, the Ministry of Public Health in Thailand, Pharco Pharmaceuticals in Egypt, and Pharmaniaga in Malaysia to jointly develop ravidasvir using this alternative pathway. Ravidasvir is an investigational NS5A inhibitor. Pharco agreed to collaborate on the development of ravidasvir and to supply generic sofosbuvir for the clinical trials of the ravidasvir-sofosbuvir combination. At this year CROI data from the HCV STORM-C-1 study was presented which aimed to assess the efficacy and safety of ravidasvir plus sofosbuvir in adults chronically infected with HCV, with or without HIV coinfection (18). STORM-C-1 was a two-stage, open-label, phase 2/3 single-arm clinical trial conducted in 13 public hospitals in Malaysia and Thailand. Participants with HCV, aged 18-69 years, without cirrhosis or with compensated cirrhosis (Child-Turcotte-Pugh class A), were eligible to participate, regardless of HCV genotype, HIV infection status or previous interferon-based HCV treatment. Once daily ravidasvir (200 mg) and sofosbuvir (400 mg) were prescribed for 12 weeks for participants without cirrhosis or 24 weeks for those with cirrhosis. The primary endpoint was sustained virological response at 12 weeks after treatment (SVR12; defined as HCV RNA < LLOQ). Between September 2016, and September 2020, 603 participants were enrolled in STORM-C-1. Of these, 296 (49%) had genotype 3 infection, 162 (27%) had genotype 1a, 81 (13%) had genotype 1b, 61 (10%) had genotype 6 and 3 (<1%) had genotype 2. 238 (39%) had compensated cirrhosis, 192 (32%) had HIV co-infection, and 120 (20%) had received previous interferon-based treatment. SVR12 was achieved by 583/602 (96.8%; 95% CI 95.1- 98.1) participants. Overall SVR12 rate in per protocol set (n=580): 98.1% (95% CI: 96.6% to 99.0%). There were no significant drug-drug interactions with anti-retroviral therapies. Most common adverse events were pyrexia (8%), urinary tract infection (6%), cough (6%), dizziness (5%), and headache (5%). One treatment emergent serious adverse event of acute kidney injury was assessed as possibly related to study treatment (sofosbuvir). Otherwise, tolerability was very good. The SVR rates overall, as well as for various subgroups are shown in figure 10.
Figure 10: Sustained virological response at 12 weeks post-treatment - Full analysis set (n=602).
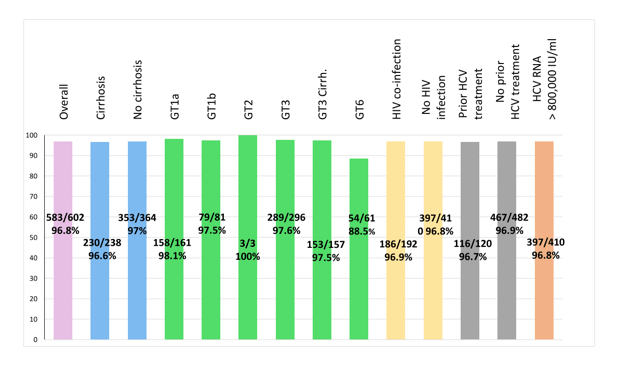
Efficacy was maintained throughout all subgroups with the exception of genotype 6 patients were SVR rate was somewhat lower with 88.5%. In the poster discussion the authors highlighted that most patients with GT6 have RAVs on positions 28V and 93S, known as risk factors for virological failure. Therefore, more data is needed for the various HCV genotype 6 subtypes.
In summary, ravidasvir + sofosbuvir was well tolerated with excellent safety and efficacy in chronic HCV infection, including in difficult-to-treat populations (GT3, cirrhosis, prior HCV treatment, HIV co-infection).
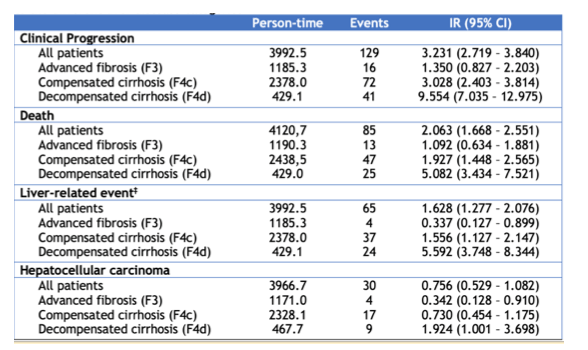
Direct-acting antivirals (DAAs) are highly successful in HIV/HCV-coinfected patients with advanced fibrosis (F3) or cirrhosis (F4), but little is known about their impact on clinical events. At CROI 2022 a study was presented from Spain which assessed the clinical outcomes of HIV/HCV-coinfected patients with F3/F4 following a sustained viral response (SVR) following all-oral DAA therapy and tried to identify prognostic factors (10). All coinfected patients with F3/F4 with SVR following all-oral DAA therapy from 2014 to 2017 from the observational GeSIDA cohorts (Spain) were included. The censoring date was December 31, 2019. Liver disease categories were defined by liver stiffness assessed by transient elastography (TE), liver biopsy, and clinical-biological parameters. The TE cut-offs were > 9.9 and ≤ 12.5 kPa for F3, and > 12.5 kPa for F4. Decompensation (DEC) was defined by the presence of ascites, variceal bleeding, or portosystemic encephalopathy. Hepatocellular carcinoma (HCC) was defined by imaging criteria (CT & MR) defined by the EASL. The primary outcome was time from the finalization of DAA therapy to clinical progression (CP), defined as DEC, HCC, or death, whichever occurred first. A total of 1300 patients were included with a median age of 52 years; 79% males; 87% prior injection drug use; 98% on ART; 94% with undetectable HIV-RNA; median CD4+ 525 cells/mm3. Liver disease: 384 (30%) F3, 761 (59%) F4c, and 155 (12%) F4d. After a median follow-up of 40.9 (34.5 - 45.1) months, 89 patients were lost, 85 died, 65 had a new LRE (DEC or HCC), and 30 were diagnosed with HCC. The frequency and incidence rate of outcomes by liver disesase category is shown in the Table 3.
Table 3: Frequency and incidence rate rate x 100 person-years of outcomes according to the different liver disease categories.
The cumulative incidence plots for the different outcomes for the different liver disease category are shown in Figure 11.
Figure 11: Cumulative incidence plots for the different outcomes for the different liver disease categories.
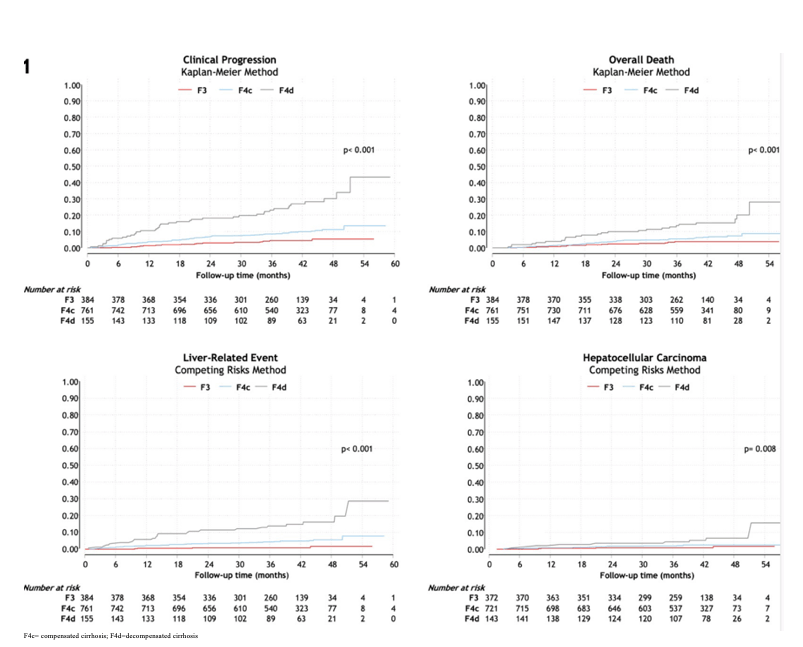
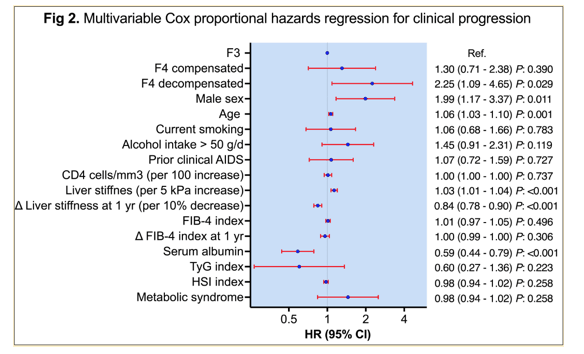
The following variables were found to be independently associated with clinical progression: F4d (vs F3) (adjusted hazard ratio [aHR] 2.25; 95%CI 1.09-4.65, P=.029), male sex (aHR 1.99; 95%CI 1.17-3.37, P=.011), age (aHR 1.06; 95%CI 1.03-1.10, P=.001), liver stiffness (LS) (per 5kpa increase) (aHR 1.03; 95%CI 1.01-1.04, P<.001), delta-LS at one year (per 10% decrease) (aHR 0.98; 95%CI 0.98-0.99, P<.001), and serum albumin (aHR 0.59; 95%CI 0.44-0.79, P<.001).
In conclusion, these study results from a large cohort of HIV/HCV coinfected patients with advanced fibrosis and SVR after DAA therapy and well controlled HIV-infection demonstrate a remaining risk for the development of liver disease associated events. The risk of clinical progression is independently associated with age, sex, liver disease severity, and changes in liver stiffness one year after finalization of therapy. The authors underline that further work should be done to develop prediction scores to inform clinical decision-making in this population group.
HIV and fatty liver disease
Nonalcoholic fatty liver disease (NAFLD) can progress to cirrhosis, hepatocellular carcinoma, and end-stage liver failure. Indeed, NAFLD has also become a major cause of liver disease including among persons with HIV. The NAFLD incidence in HIV-coinfection however still remains unclear. At this year CROI data was presented from the Multicenter AIDS Cohort Study (MACS), a prospective cohort of men who have sex with men which aimed to determine NAFLD incidence among persons with and without HIV (11). Participants of the MACS Coronary Atherosclerosis Progression Study with all the following criteria were included: two non-contrast CT scans with complete visualization of the liver and spleen, consumed <3 alcoholic drinks/day on average, and had stored cells available for PNPLA3 testing. Further inclusion criteria for the MACS Coronary Atherosclerosis Progression Study were age 40-70, no prior coronary intervention, and eGFR >60 mL/min/1.73m². The baseline CT scans were performed from 2010-2013 and follow-up scans from 2015-2017. NAFLD was defined as liver/spleen HU ratio <1.0.6. Visceral adipose tissue (VAT) was measured in one axial image obtained between the 4th and 5th lumbar vertebrae. 268 men were eligible: 173 men with HIV (MWH) and 95 men without HIV (MWOH). Main baseline characteristics were: median age 57 (IQR 53-62), 53% White non-Hispanic, 35% Black non-Hispanic. The median time between CT scans was 4.5 years (IQR 3.8-5.0). Incident NAFLD was detected in 30 men (11.1%) with an overall IR of 2.5/100 PYs (95% CI 1.8-3.6). Table 4 shows the baseline factors which were associated with incident NAFLD.
Table 4: Higher BMI and VAT at baseline but not HIV were associated with incident NAFLD.*
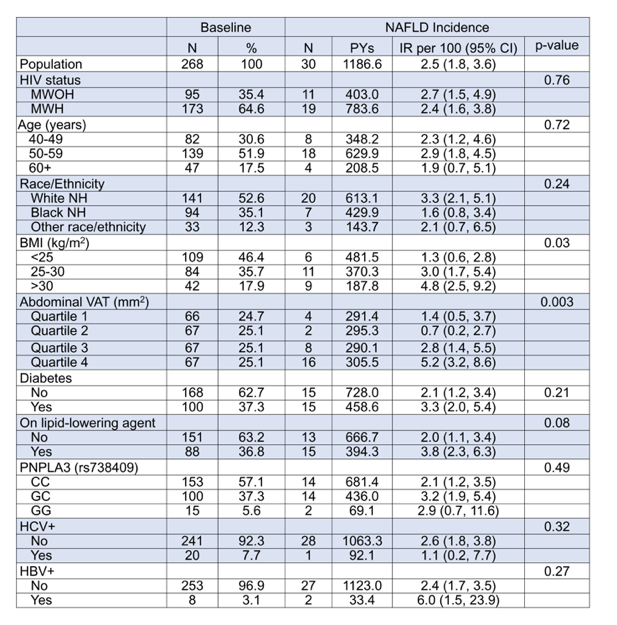
*Additional covariates not significantly associated with NAFLD incidence include: HOMA-IR, abdominal SAT, HDL, LDL, triglycerides, hypertension, undetectable HIV RNA, CD4 cell count <500 cells/mm3, current HAART, cumulative HAART, and cumulative NNRTI, NRTI, and PI use.
In summary, visceral adiposity, but not HIV infection, was associated with incident NAFLD as determined by serial non-contrast CT scans. Although MWH were not at higher risk of NAFLD than MWOH, the incidence of NAFLD among MWH in the study (2.4/100 PYs) was higher than the incidence rates previously published for hepatitis B virus (1.5/100 PYs) and hepatitis C virus (0.4/100 PYs) among MWH in the larger MACS cohort. Moreover, the high incidence of ≥ moderate steatosis suggests that NAFLD will continue to increase as a cause of liver disease in persons with HIV.
Given the impact of new antiretroviral therapy (ART), including integrase strand inhibitors (InSTI) and tenofovir alafenamide (TAF), on weight and metabolic parameters, their potential
contribution to the development of liver steatosis is of concern. Indeed, more recently it was published that steatosis can even develop in non-obese BMI in patients with HIV monoinfection under cART (19). While TAF and INSTI appeared to increase the risk of progression of steatosis, TDF was found to be independently associated with a lower risk of a clinically significant weight gain and thereby, might slow down development and progression of steatosis. At this year CROI new results from the Swiss Cohort study (12) were presented which evaluated the determinants of liver steatosis among people living with HIV (PLWH) in a single center of the Swiss HIV Cohort Study (SHCS), and described the proportion of individuals with metabolic dysfunction-associated fatty liver disease (MAFLD), according to the 2020 definition (20). PLWH from the Bern University Hospital were enrolled between November 2019 and August 2021. Individuals with active or past viral hepatitis co-infection and pregnant women were excluded. All participants were asked to undergo liver stiffness measurement (LSM) and controlled attenuation parameter (CAP) using transient elastography (TE). Liver steatosis was defined as follows: S1 (mild steatosis) if CAP 248-267dB/m, S2 (moderate steatosis) if CAP 268-279dB/m, and S3 (severe steatosis) if CAP ≥280dB/m. Liver fibrosis was categorized according to Metavir equivalent stage: F0-1 (no or mild fibrosis) if LSM <7.1kPa, F2-3 (significant fibrosis) if LSM 7.1-11kPa, and F4 (cirrhosis) if LSM ≥ 11.1kPa 2. Multivariable logistic regression was used to explore factors associated with steatosis. Table 5 summarizes the main baseline characteristics by BMI category.
Table 5: Baseline characteristics, stratified by BMI category
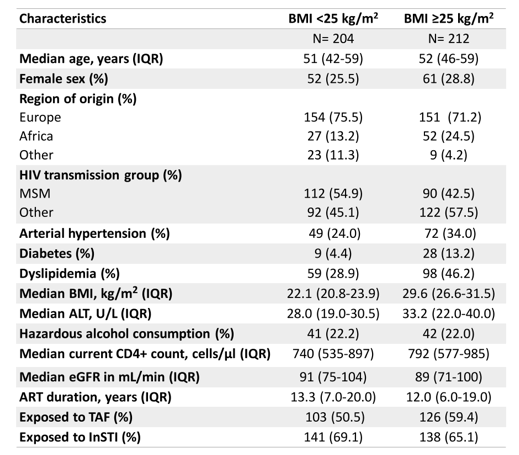
Of 480 eligible individuals, 416 (86.7%) agreed to participate and had a reliable TE measurement. Cardio-metabolic risk factors were more common in patients with a BMI ≥25 kg/m2. A similar proportion of participants was exposed to InSTI in both groups (69% and 65%, respectively). Among individuals with a BMI <25 kg/m2, 50.5% were on a TAF-containing regimen, whereas this proportion was 59.4% among patients with a BMI ≥25 kg/m2. Overall, 212 (51.0%) participants had liver steatosis, including 143 with severe steatosis. The proportion of individuals with liver steatosis was 69.8% in patients with a BMI ≥25 kg/m2 and 31.4% in individuals with a BMI <25 kg/m2. Of all participants, 179 (43.0%) met the criteria for MAFLD. In multivariable analyses, patients were more likely to have liver steatosis if they were ≥50 years, of European origin, or had a BMI ≥25 kg/m2 (see figure 12). Whereas no association was found between InSTI exposure and liver steatosis. In contrast, participants currently on a TAF-containing regimen were more likely to have steatosis than those not on TAF (see figure 12). Interestingly, the latter association did not depend on the time spent on a TAF-containing regimen. When weight increase of 10% was included as a further risk factor to the multivariable model, both weight gain (aOR, 3.12; 95% CI, 1.42-6.83) and exposure to TAF (aOR, 1.65; 95% CI, 1.03-2.65) remained significantly associated with liver steatosis.
Figure 12: Multivariate analysis of factors associated with liver steatosis
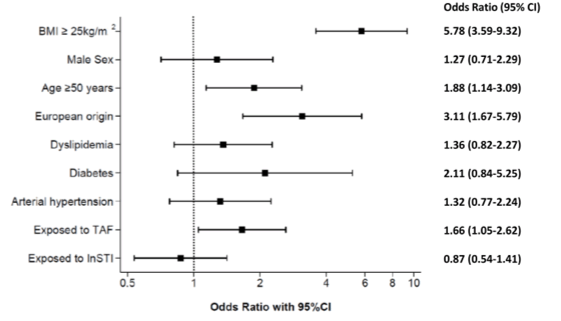
Abbreviations: BMI, body mass index, TAF; tenofovir alafenamide; InSTI, integrase-strand transfer inhibitor; OR, odds ratio; CI, confidence interval
In conclusion, this study suggests a high prevalence of liver steatosis among PLWH on ART in Switzerland. In addition to well-established risk factors such as age, ethnicity and obesity, the use of TAF was significantly associated with hepatic steatosis. Overall, MAFLD was diagnosed in 43% of participants, including in 17% of individuals with a BMI <25 kg/m2and liver steatosis. These data underline that potentially with some of the newer modern antiretroviral therapies there may be an increased risk for development of fatty liver disease which warrants closer monitoring of liver fibrosis and steatosis over time in PLWH under ART.
Interesting work was also presented around the impact of NAFLD on the prognosis of PLWH. Within a Spanish cohort study, the influence of non-alcoholic steatohepatitis (NASH) with fibrosis on the overall mortality in PLWH was evaluated (13). PLWH followed in three Spanish centers were included in a prospective cohort at the date when a vibration-controlled transient elastography (VCTE) evaluation, including controlled attenuation parameter (CAP) measurement was conducted for the first time. Clinical visits were scheduled, at least, every 6 months. Survival data was recorded, and the causes of death were centrally monitored. The risk of all-cause of death were evaluated applying time-to-event analyses. NAFLD was defined as steatosis (CAP ≥248 dB/m) without any other liver disease. Cirrhosis was defined as liver stiffness measurement (LSM) ≥10.3 kPa for NAFDL. The FibroScan-AST (FAST) score, which includes AST, CAP and liver stiffness measurement (LSM) was calculated. 1810 PLWH were included in the cohort and followed for a median of 74 (23-102) months. There were 134 (7.4%) deaths. The main causes of death were: Cancer, 38 (28%), liver-related, 36 (27%); AIDS, 15 (11%); cardiovascular, 12 (9%); other, 33 (25%) (Other was: Non-AIDS infections: 10 (7.5%); overdose: 8 (6%); chronic lung disease: 7 (5.2%); accidental: 1 (0,7%); unknown cause: 7 (5.2%)). Steatosis was not associated with overall mortality. However, for PLWH with NAFLD, the higher the LSM, the lower the probability of survival (see figure 12). FAST score ≥0.67 was also associated with a lower likelihood of survival in PLWH with NAFLD (see figure 13).
Figure 13: Survival curves overall and for PLWH without concomitant causes of liver disease according to liver stiffness and FAST score.
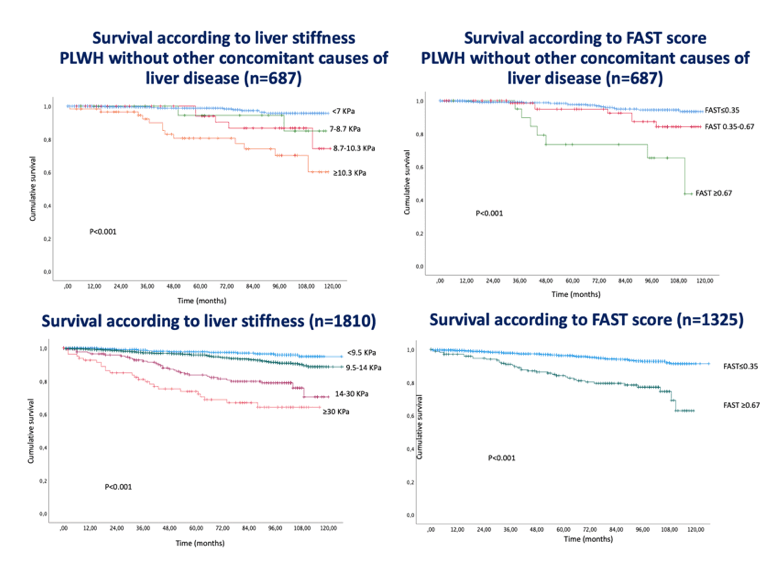
In conclusion, among PLWH, the presence of NAFLD, as a whole, is not associated with survival. However, for PLWH without concomitant liver diseases other than NAFLD, liver fibrosis increases the risk of death. Liver stiffness and the FAST score were similar predictors of survival in this setting.
Unfortunately, the treatment options for fatty liver disease are still limited. Interesting data at CROI were presented for cannabis and its potential effect on hepatic steatosis (14). Cannabis affects many gastrointestinal processes via the endocannabinoid system, which plays an important role in fatty liver diseases. The data on the impact of cannabis use on liver steatosis in people living with HIV and/or HCV infections so far has been conflicting. At CROI 2022 a cross-sectional analysis of the Miami Adult Studies on HIV (MASH) cohort study was presented which assessed the relationship between the use of cannabis and hepatic steatosis in a cohort of people living with and without HIV and HCV (14). The use of cannabis in the past 30 days was self-reported. Hepatic steatosis (>5%) was assessed with magnetic resonance-determined proton density fat-fraction (MRI-PDFF). There were 467 participants included into the study of whom 38.5% were HIV mono-infected, 13.7% were HCV mono-infected, and 12.4% were HIV/HCV co-infected. All people living with HIV were on antiretroviral treatment (ART). Sixty-eight participants (14.6%) had hepatic steatosis. Multivariate logistic regression showed that BMI (kg/m2; adjusted odds ratio [aOR] 1.11, 95% CI 1.06-1.17, P <0.001) and cannabis use (aOR 0.48, 95% CI 0.26-0.91, P=0.024) were significant independent predictors of hepatic steatosis after adjustment for covariates (race/ethnicity, BMI, smoking, and cocaine use). Most interestingly, cannabis use was associated with reduced risk of hepatic steatosis in both obese (relative risk [RR] 0.47, 95% CI 0.22-0.98, P=0.044) and non-obese (RR 0.37, 95% CI 0.14-0.94, P=0.038) participants. The use of cannabis was associated with lower odds for hepatic steatosis in the HIV/HCV-uninfected and HIV mono-infected groups, but not in HCV-infected groups (see Table 6). BMI increased the risk of liver steatosis in all groups except in the HCV mono-infected group (see table 6).
Table 6: Bivariate logistic regression for hepatic steatosis (> 5% fat) stratified by HIV/HCV status.
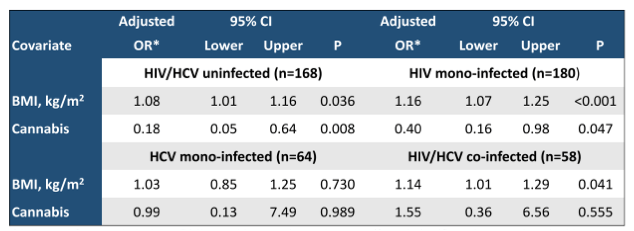
*Adjusted for race/ethnicity, BMI, smoking, and cocaine use (found significantly associated with hepatic steatosis in univariate analysis).
In summary, the use of cannabis may reduce the risk of hepatic steatosis in HIV/HCV-uninfected and HIV mono-infected, but not in HCV-infected individuals. The analyses showed this association both in obese and non-obese participants. Further studies are needed to explore and help better understand the biological mechanisms for the beneficial effect of cannabis use observed in this study.
Summary
• HBV revaccination with three double doses of HBV vaccine achieves higher serological responses than with three standard-doses of HBV vaccine among MSM who were born in the era of universal neonatal HBV vaccination. Reassuringly, virologically suppressed PLWH with CD4-counts >500 cells/mm³ show similar responses after revaccination as healthy MSM.
• PWH who are unvaccinated for hepatitis B are at higher risk for new hepatitis B infections. A particular high risk was found among those individuals with hepatitis C. In conclusion, this study strongly recommends HBV vaccination in HBV seronegative PLWH even if they are on dual active HIV/HBV antiviral therapy.
• A nanoformulated TFV prodrug suppresses HBV DNA in humanized and transgenic mice for three months. These data suggest that a long-acting tenofovir formulation for treatment of HBV infection can be developed.
• The U.S. is currently not on track to achieve the WHO elimination targets pre-pandemic and has fallen further behind during the COVID-19 pandemic. Increased efforts including scale-up of HCV diagnosis, treatment, and prevention are needed to overcome this unfavorable development.
• Pooled analysis from Australia and selected Western European countries demonstrates that following broad access to DAA therapy these countries are on track to meet the WHO elimination incidence target for PLHIV in care by 2030.
• The German NoCo cohort demonstrates stable HCV incidence rates despite a broad use of DAAs up to 2019. In 2021, however, micro-elimination goals are met due to a drop in HCV incidence, possibly due to behavior changes related to the SARS-CoV-2 pandemic and associated containment measures or because of a declining HCV reservoir over time with many MSM successfully treated with DAAs. These results emphasize that HCV elimination in MSM is achievable.
• With regular HCV testing and improved access to antiviral treatments, particularly highly effective direct-acting antivirals (DAAs) in Taiwan, the incidence rate and prevalence of HCV viremia have declined by 80% among people living with HIV between 2011 and 2021.
• Risk-based HCV screening is insensitive for HCV detection within the general obstetric population. In contrast universal HCV screening of pregnant people does not only ensure that the pregnant person is linked to treatment, but also enables detection of all cases of perinatal HCV transmission.
• SOF/VEL/VOX is highly effective for HCV treatment in PLHIV previously failing to DAA regimens. Effectiveness was confirmed across all genotypes and in the presence of cirrhosis.
• Ravidasvir + sofosbuvir was well tolerated with excellent safety and efficacy in chronic HCV infection, including in difficult-to-treat populations (GT3, cirrhosis, prior HCV treatment, HIV co-infection).
• The risk of clinical progression to liver disease associated events following DAA-induced SVR in HIV/HCV coinfected subjects with advanced fibrosis is independently associated with age, sex, liver disease severity, and changes in liver stiffness one year after finalization of HCV therapy.
• Within the MACS Coronary Atherosclerosis Progression Study the incidence rate of CT-measured NAFLD was high at 2.5/100 PYs. Higher BMI and visceral adiposity, but not HIV, were associated with incident NAFLD.
• A high prevalence of liver steatosis can be found among PLWH on ART in Switzerland. In addition to well-established risk factors such as age, ethnicity and obesity, the use of TAF was significantly associated with hepatic steatosis.
• Among PLWH without concomitant causes of liver disease other than NAFLD, liver stiffness as well as FAST score predict overall survival.
• Cannabis use may reduce the risk of hepatic steatosis in HIV/HCV-uninfected and HIV mono-infected, but not in HCV-infected individuals. Further studies are needed to help better understand the biological mechanisms for the beneficial effect of cannabis use observed in this study.
References
1. Ortega REF et al. Modelling impact of COVID-19 pandemic on hepatitis C virus elimination in the US. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 72
2. Van Santen DK et al. Effect of direct-acting antivirals on HCV incidence among people living with HIV. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 73
3. Chappell C et al. Uprising circulation of HBV-complex profiles with HBsAG vaccine-escape mutations. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2020, abstract 27
4. Huang C et al. Randomized Trial of HBV Revaccination in MSM Born in the Neonatal Vaccination Era. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 544
5. Jain MK et al. New Hepatitis B infection among HIV patients: Who is at risk? 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 548
6. Das S et al. A nanoformulated TFV prodrug suppresses HBV DNA in humanized and transgenic mice for three months. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 545
7. Ingiliz P et al. The impact of COVID-19 on HCV microelimination in MSM in Germany. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 539
8. HO SY et al. Progression towards HCV microelimination among HIV-positive patients in Taiwan. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 538
9. Carbonero LM et al. Treatment with SOF/VEL/VOX in HIV-HCV coinfected patients previously exposed to DAAs. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 529
10. Berenguer J et al. Impact of SVR with DAAs in coinfected patients with advanced fibrosis/cirrhosis. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 531
11. Price J et al. High incident rate of CT-measured NAFLD in men with and without HIV infection. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 523
12. Riebensahm C et al. Determinants of liver steatosis in people living with HIV on antiretroviral therapy. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 524
13. Macias J et al. Impact of NASH on the survival of people living with HIV. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 518
14. Diaz-Martinez J et al. Cannabis use and hepatic steatosis by MRI-PDFF. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 525
15. Jain MK et al. Low rates of hepatitis B vaccination among people with HIV. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 546
16. Heuft MM, et al. Protective effect of hepatitis B virus-active antiretroviral therapy against primary hepatitis B virus infection. AIDS. 2014;28:999-1005.
17. Kusejko K et al. A systematic re-screening for hepatitis C RNA among men who have sex with men living with HIV in the Swiss HIV Cohort Study two years following a nation-wide elimination program reveals a sustainable effect towards hepatitis C elimination. EACS 2021; OS2/3
18. Tan SS et al. Efficacy & safety of ravidasvir + sofosbuvir in hepatitis C: STORM-C-1 final results. 29th Virtual Conference on Retroviruses and Opportunistic Infections, February 12-16, 2022, abstract 528
19. Bischoff J et al. Stratifying the risk of NAFLD in patients with HIV under combination antiretroviral therapy (cART). EClinicalMedicine. 2021 Sep 5;40:101116
20. Eslam M, et al. A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement. J Hepatol 2020; 73(1):202-209.
|
| |
|
 |
 |
|
|