 |
 |
 |
| |
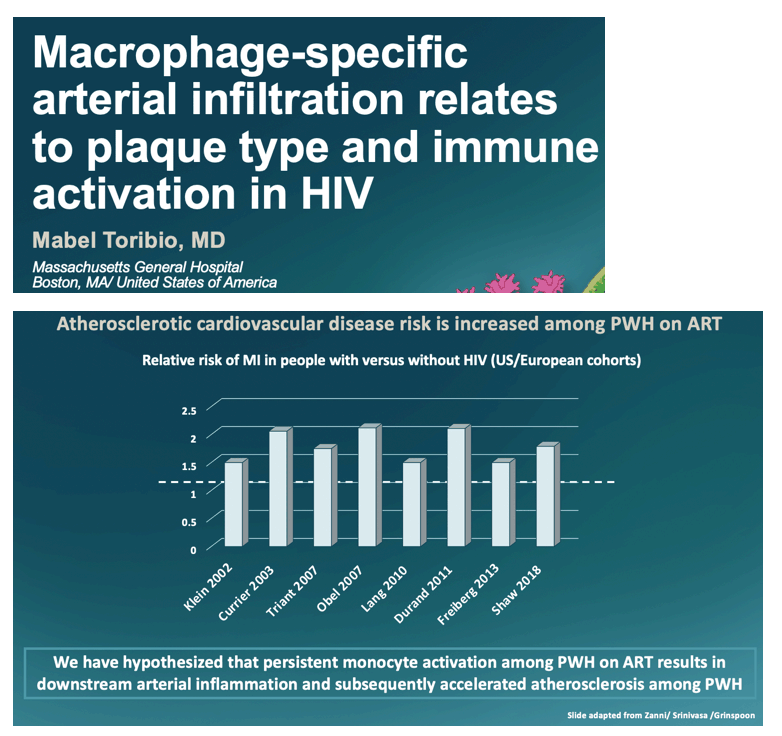
Macrophage-specific arterial infiltration relates to plaque type and immune activation in HIV
|
| |
| |
Mabel Toribio, MD
Massachusetts General Hospital
Boston, MA/ United States of America
program abstract
Background:
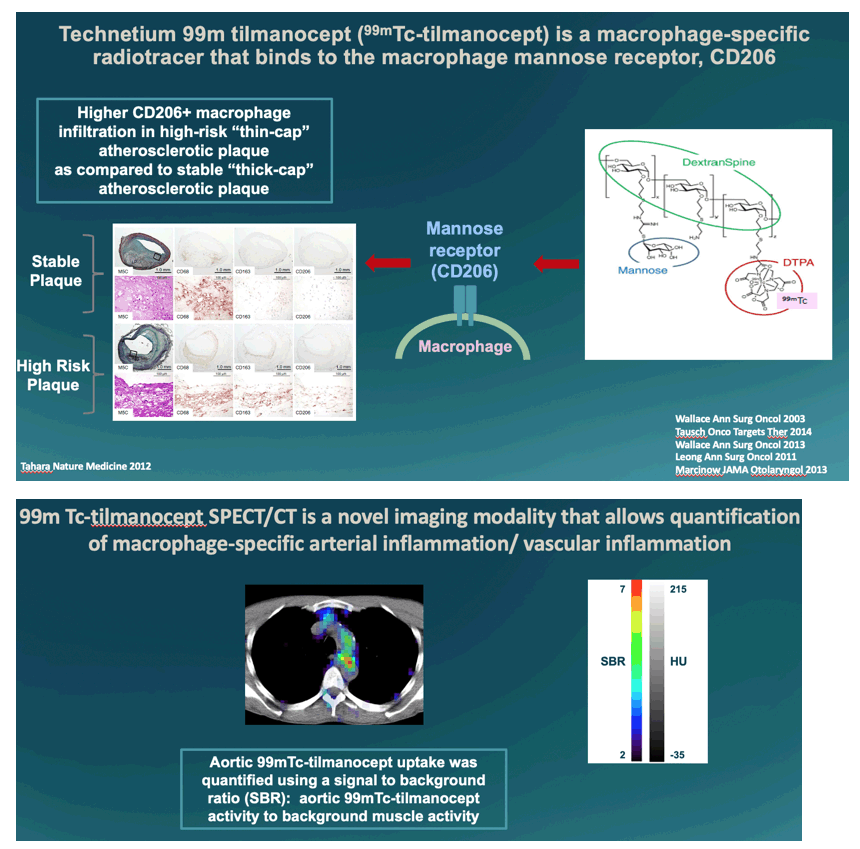
Persistent immune activation and downstream macrophage-specific arterial infiltration are thought to contribute to heightened atherosclerotic cardiovascular disease (ASCVD) risk among people with HIV (PWH) on ART. We applied a novel macrophage-specific imaging modality to investigate macrophage-specific infiltration among participants with vs without HIV in relation to atherosclerotic plaque and immune activation.
Methods:
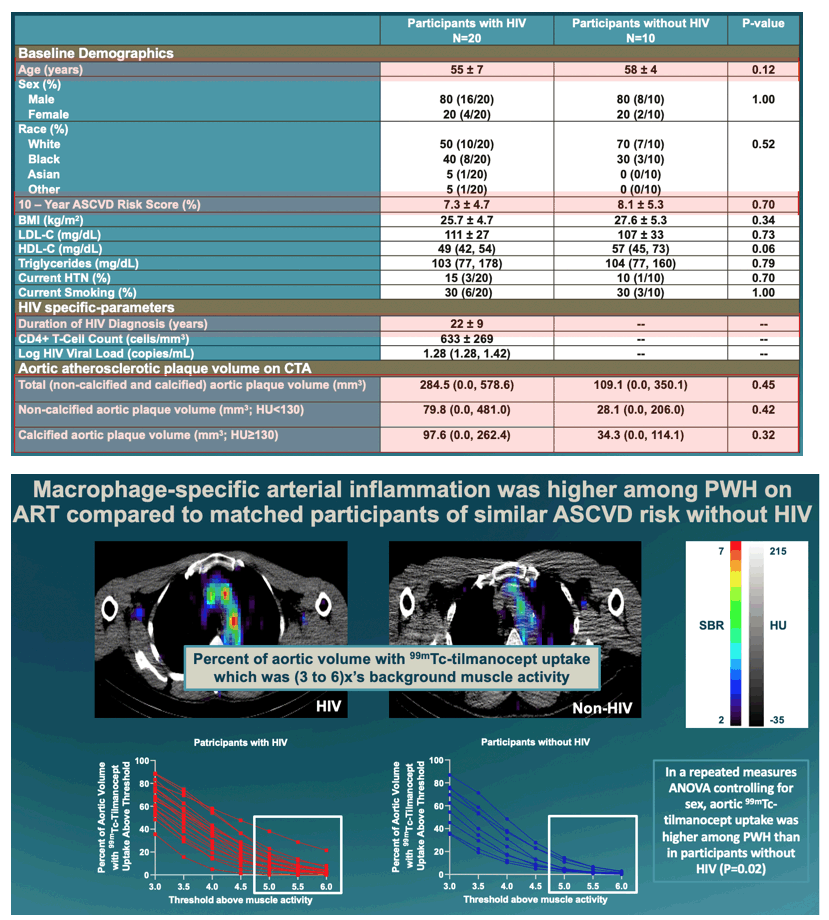
Twenty PWH on ART and 10 participants without HIV underwent systemic administration of the CD206 macrophage-specific radiotracer, 99mTc-tilmanocept, followed by SPECT/CT imaging to assess arterial inflammation. Participants were ≥18 yrs and without a history of symptomatic ASCVD. The volume of aortic tilmanocept uptake that was 3-6x background muscle activity (signal to background ratio, SBR) was measured. Aortic plaque volumes [total, non-calcified (Hounsfield units (HU) <130 and HU <300), and calcified plaque (HU≥130 and HU≥300)] were quantified using cardiac CT.
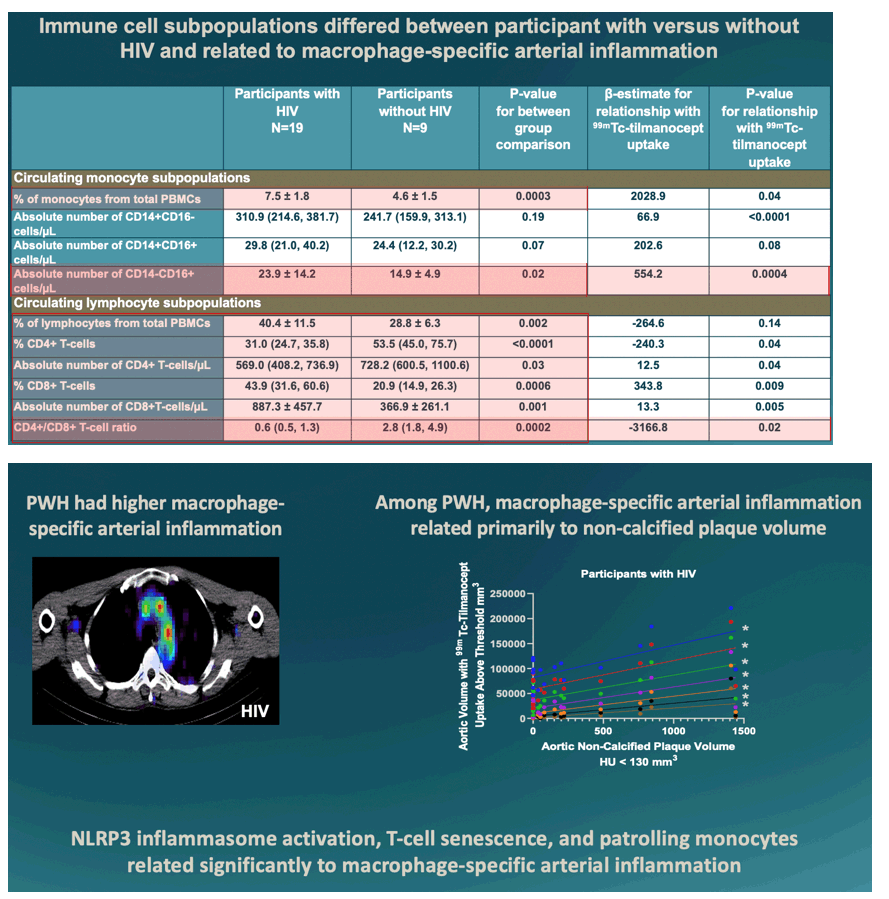
Results:
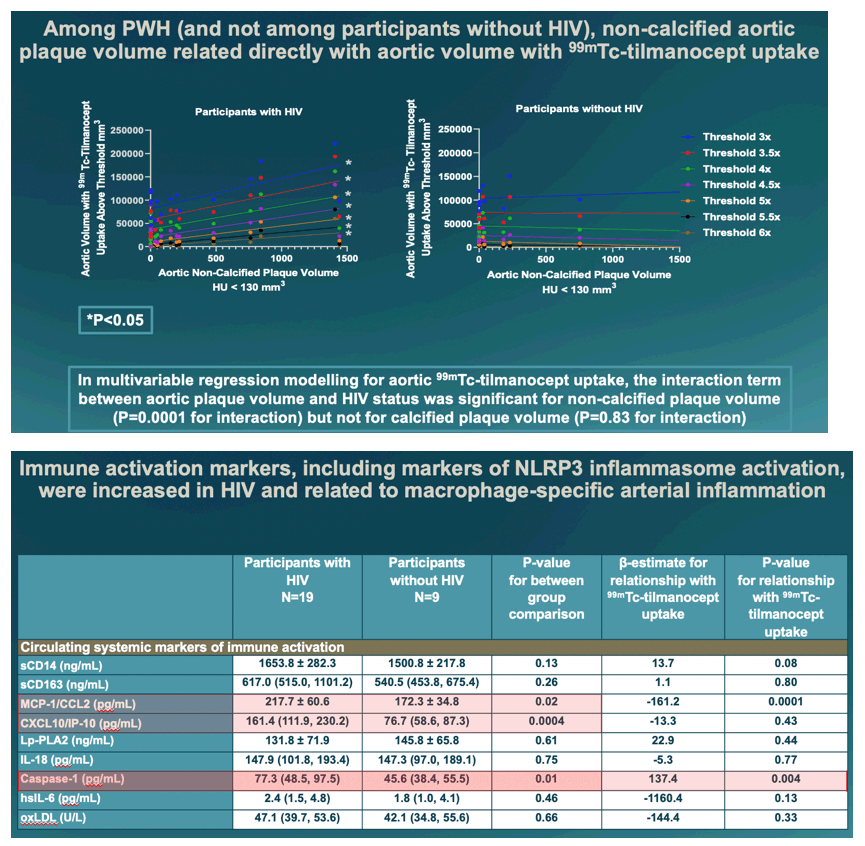
Participants with vs without HIV were similar in age (55.1 vs 58.4 yrs, P=0.12) and 10-yr ASCVD risk (7.3 vs. 8.1, P=0.70). Total, non-calcified, and calcified aortic plaque volume did not differ significantly between groups. Systemic markers of immune activation (caspase-1: P=0.01, MCP-1: P=0.02, and CXCL10: P=0.0004) and non-classical/homing monocytes (CD14-CD16+: P=0.02) were higher among PWH. Aortic tilmanocept uptake was higher across different uptake thresholds among PWH (P=0.03; Fig 1a-b). There was a significant interaction between HIV status and plaque volume in relation to arterial inflammation for non-calcified plaque (P=0.0001, Fig 1c) but not with calcified plaque (P=0.83) with significant relationships between non-calcified aortic plaque volume with each tilmanocept uptake threshold for PWH but not participants without HIV (Fig 1d). Markers of immune activation (sCD163 P=0.045 and caspase-1, P=0.02), immune cell subpopulations (CD14+CD16- P=0.0001, CD14+CD16+, P=0.02 and CD8+ T-cells P=0.01), and CD4+/CD8+ ratio (P=0.02) related to aortic tilmanocept uptake volume among PWH but not among participants without HIV.
Conclusion:
These data applying a novel imaging modality provide unique evidence of macrophage-specific arterial inflammation among PWH on ART, which relates specifically to non-calcified plaque. Additionally, we characterize key immune pathways relating to increased arterial inflammation of relevance to identifying novel immunomodulatory therapies for CVD reduction.


|
| |
|
 |
 |
|
|