 |
 |
 |
| |
Obesity Risk Higher With DTG Than Other Regimens
in 3 Big African Trials - ADVANCE, NAMSAL, VISEND
|
| |
| |
HIV Drug Therapy Glasgow, October 23-26, 2022
Mark Mascolini
Analysis of data through 96 to 192 weeks in three African trials randomizing HIV-positive people to dolutegravir (DTG) versus efavirenz (EFV) or DTG versus a protease inhibitor (PI) found higher rates of obesity (body mass index above 30 kg/m2) with DTG, especially when this integrase inhibitor was combined with tenofovir alafenamide/emtricitabine TAF/FTC [1].
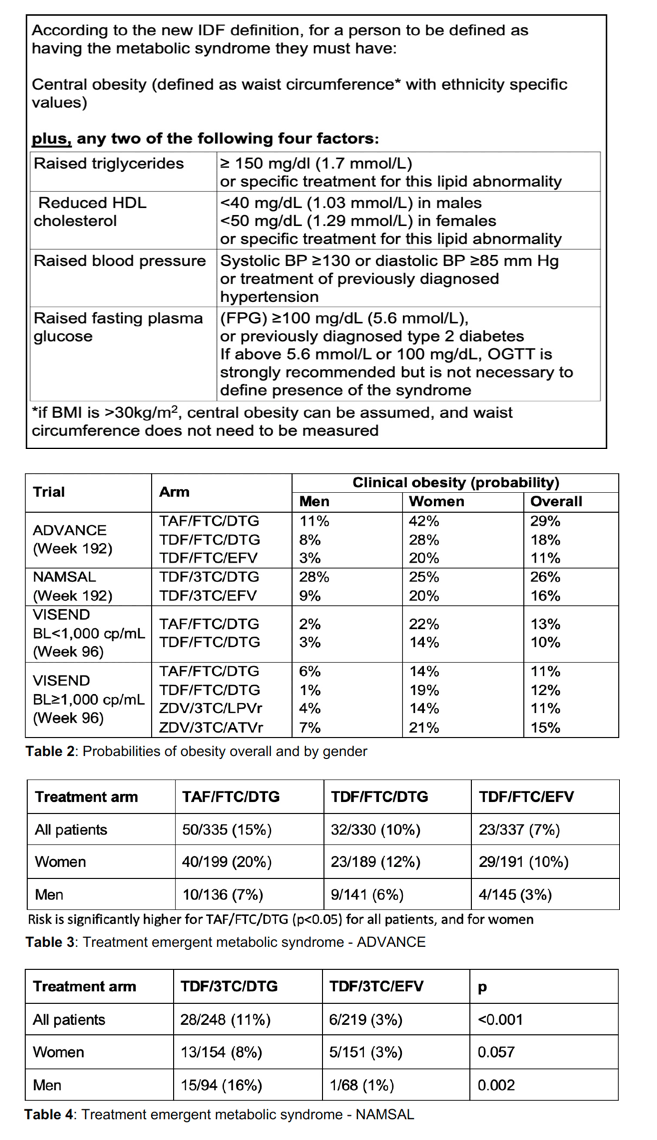
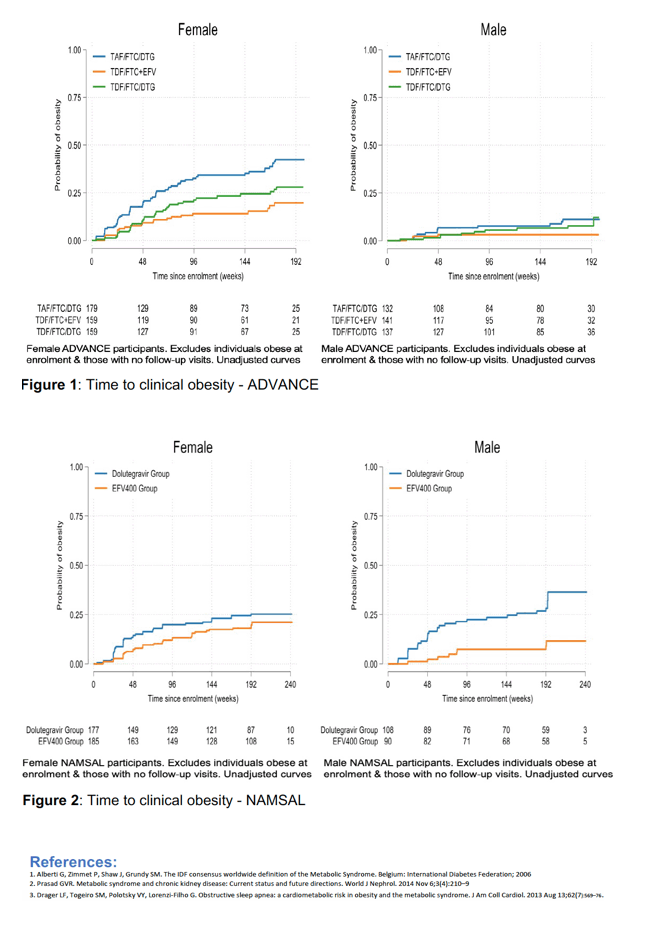
The South African ADVANCE trial randomized 1053 antiretroviral-naive people to DTG/TAF/FTC, DTG/TDF/FTC, or EFV/TDF/FTC [2]. At 192 weeks, proportions of participants with treatment-emergent obesity with those three regimens were 29%, 18%, and 11%, mostly because of high obesity rates among women: 42%, 28%, and 20%.
In Cameroon the NAMSAL trial randomized 613 previously untreated people to DTG/TDF/3TC or EFV/TDF/3TC [3]. At week 192 proportions with new obesity for those two regimens were 26% and 16%, but in this trial DTG was associated with obesity among both men (28%) and women (25%).
In Zambia the VISEND trial enrolled 1201 nonnucleoside-treated people and randomized them to DTG/TAF/FTC or DTG/TDF/FTC if their baseline viral load lay below 1000 copies. After 96 weeks proportions with obesity were 13% with DTG/TAF/3TC and 10% with DTG/TDF/FTC, with higher obesity rates in women for those two regimens-22% and 14%. People with a baseline viral load above 1000 copies got randomized to DTG/TAF/FTC, DTG/TDF/FTC, LPV/r/ZDV/3TC, or ATV/r/ZDV/3TC. For those four regimens 96-week treatment-emergent obesity rates varied less by regimen: 11%, 12%, 11%, and 15% overall, and 14%, 19%, 14%, and 21% for women.
By Cox proportional hazards models, DTG/TAF/FTC predicted obesity in ADVANCE (P < 0.001) and in the VISEND group starting treatment with a viral load below 1000 copies (P = 0.002). DTG/TDF/3TC predicted obesity in NAMSAL (P < 0.001).
In ADVANCE risk of metabolic syndrome was significantly higher with DTG/TAF/FTC for all participants (P < 0.05) and for women (P < 0.05). In NAMSAL treatment-emergent metabolic syndrome proved significantly more frequent with DTG/TDF/3TC for all participants (P < 0.001) and for men (P = 0.002), and nearly significantly more frequent for women (P = 0.057).
Researchers who ran these trials cautioned that consequences of obesity, like diabetes and myocardial infarction, must be considered when starting or switching to DTG/TAF/FTC, especially in people who are already overweight.
References
1. Venter F, Sokhela S, Bosch B, et al. Long-term risks of clinical obesity in the ADVANCE, NAMSAL and VISEND trials. HIV Drug Therapy Glasgow, October 23-26, 2022.
2. Venter WDF, Sokhela S, Simmons B, et al. Dolutegravir with emtricitabine and tenofovir alafenamide or tenofovir disoproxil fumarate versus efavirenz, emtricitabine, and tenofovir disoproxil fumarate for initial treatment of HIV-1 infection (ADVANCE): week 96 results from a randomised, phase 3, non-inferiority trial. Lancet HIV. 2020;e666-e676. doi: 10.1016/S2352-3018(20)30241-1.
https://www.thelancet.com/journals/lanhiv/article/PIIS2352-3018(20)30241-1/fulltext
3. Calmy A, Tovar Sanchez T, Kouanfack C, Dolutegravir-based and low-dose efavirenz-based regimen for the initial treatment of HIV-1 infection (NAMSAL): week 96 results from a two-group, multicentre, randomised, open label, phase 3 non-inferiority trial in Cameroon. New Antiretroviral and Monitoring Strategies in HIV-infected Adults in Low-Income Countries (NAMSAL) ANRS 12313 Study Group. Lancet HIV. 2020;7:e677-e687. doi: 10.1016/S2352-3018(20)30238-1.
https://www.thelancet.com/journals/lanhiv/article/PIIS2352-3018(20)30238-1/fulltext
4. Clinical Care Options HIV. VISEND: Switching from NNRTI-based to PI-based ART vs DTG + recycled NRTIs. Source: Conference Coverage of CROI 2022: Clinical Impact of New HIV Data. February 13-16, 2022; Virtual.
https://www.clinicaloptions.com/hiv/conference-coverage/2022/retroviruses-2022/clinical-impact-of-new-hiv-data/capsule-summary-slidesets/135

|
| |
|
 |
 |
|
|