 |
 |
 |
| |
Alcohol, Smoking, Drugs, Liver Disease-But Not TDF-Predict Fracture With HIV
|
| |
| |
AIDS 2022, July 29-August 2, Montreal
Mark Mascolini
Taking tenofovir disoproxil fumarate (TDF) did not predict fracture in a study of 6508 antiretroviral-treated people in the Kaiser Permanente healthcare system, but alcohol and drug use, smoking, chronic liver disease, and of course older age did foretell broken bones [1]. Neither gender nor race or ethnicity predicted fracture.
The investigators noted that an aging population with HIV faces both classic fracture risk factors and HIV-specific risks including inflammation, reconstitution syndrome, low CD4 count, and long-term antiretroviral therapy. A recent meta-analysis determine that people with HIV have a 50% higher fracture incidence than the general population and several independent risk factors: older age, smoking, HIV/HCV coinfection, and osteoporosis [2].
Research firmly links TDF use to lower bone mineral density, but whether that means TDF takers run a higher risk of breaking bones remained unclear. One study in US veterans did tie cumulative use of TDF (and lopinavir/ritonavir) to osteoporotic fracture [3], but the recent meta-analysis did not [2].
The retrospective Kaiser study covered the years 2000 through 2016 and involved Kaiser members in northern and southern California, and in Maryland, Virginia, and Washington, DC. Participants in this analysis had to be 40 or older and had to start a new antiretroviral regimen between January 1, 2000 and September 1, 2015. They could have antiretroviral experience when they started this regimen. The researchers defined incident fracture by ICD-9 codes indicating fracture after antiretroviral therapy began. They counted any fracture except trauma-related fractures.
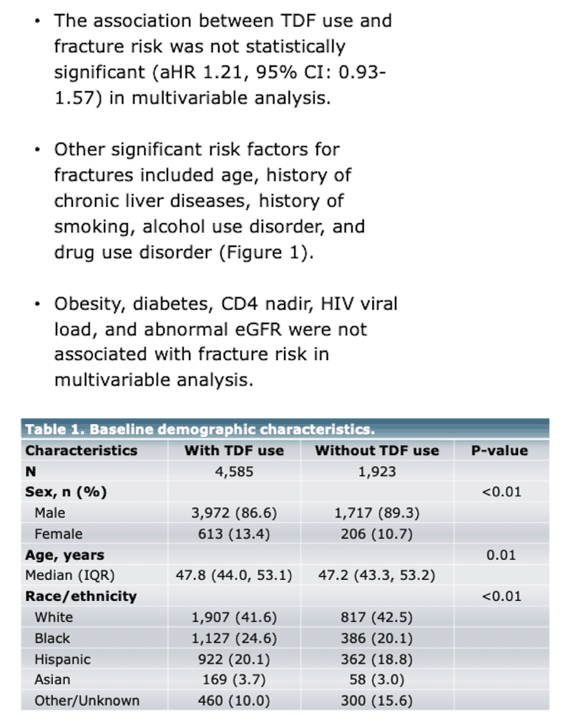
The analysis included 6508 Kaiser Permanente clients, 87% of them men and 52% men who have sex with men. A sizeable majority, 70.5%, started a TDF-containing regimen. The proportion of men was lower in the TDF group (86.6% vs 89.3%, P < 0.01), and TDF takers were slightly but significantly older (median 47.8 vs 47.2, P = 0.01). The TDF group had a lower proportion of whites (41.6% vs 42.5%), a higher proportion of blacks (24.6% vs 20.1%), a higher proportion of Hispanics (20.1% vs 18.8%), and a higher proportion of Asians (3.7% vs 3.0%) (P < 0.01).
During a total follow-up of 27,646 person-years (individual median 3 person-years), 232 fractures occurred in people who used TDF for an incidence of 14.0 per 1000 person-years (95% confidence interval [CI] 13.9 to 14.2), while 128 fractures occurred in people who did not use TDF for an incidence of 11.5 per 1000 person-years (95% CI 11.3 to 11.7).
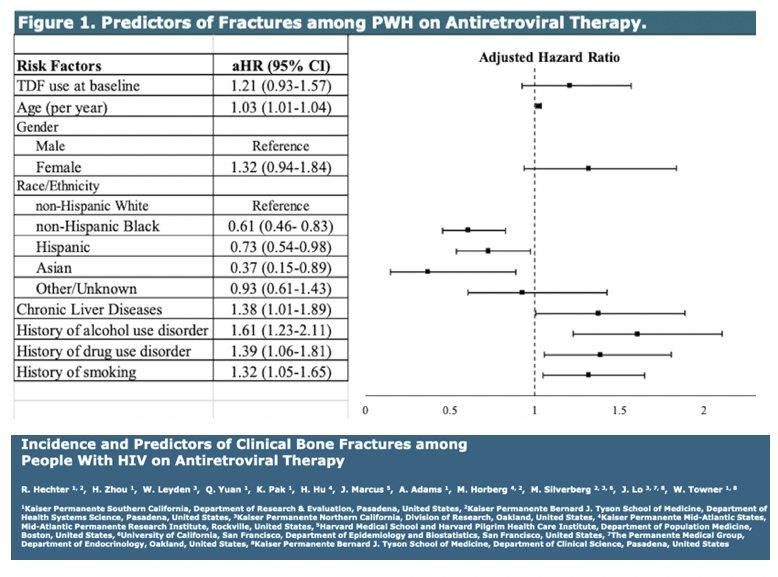
A multivariate Cox proportional hazards regression model determined that the association between TDF use and fracture was not statistically significant (adjusted hazard ratio [aHR] 1.21 95% CI 0.93 to 1.57). Four clinical or behavioral variables did independently heighten fracture risk:
- Chronic liver disease: aHR 1.38, 95% CI 1.01 to 1.89
- History of alcohol use disorder: aHR 1.61, 95% CI 1.23 to 2.11
- History of drug use disorder: aHR 1.39, 95% CI 1.06 to 1.81
- History of smoking: aHR 1.32, 95% CI 1.05 to 1.65
Obesity, diabetes, abnormal estimated glomerular filtration rate (a kidney function marker), lowest-ever CD4 count, HIV viral load, and gender did not affect fracture risk. But as in previous studies, age and race or ethnicity did:
- Every additional year of age: aHR 1.03, 95% CI 1.01 to 1.04
- Black vs white: aHR 0.61, 95% CI 0.46 to 0.83 (lower risk)
- Hispanic vs white: aHR 0.73, 95% CI 0.54 to 0.98 (lower risk)
- Asian vs white: aHR 0.37, 95% CI 0.15 to 0.89 (lower risk)
The Kaiser investigators urged clinicians to address the clinical and behavioral factors that boosted fracture risk by screening for and treating alcohol and drug use, helping people quit smoking, and treating chronic liver disease.
References
1. Hechter R, Zhou H, Leyden W, et al. Incidence and predictors of clinical bone fractures among people with HIV on antiretroviral therapy. AIDS 2022, July 29-August 2, Montreal. Abstract EPC089.
2. Pramukti I, Lindayani L, Chen YC, et al. Bone fracture among people living with HIV: A systematic review and meta-regression of prevalence, incidence, and risk factors. PLoS One. 2020;15(6):e0233501. doi: 10.1371/journal.pone.0233501. https://pubmed.ncbi.nlm.nih.gov/32497105/
The pooled estimated prevalence of fracture among PLWH was 6.6% (95% CI: 3.8-11.1) with pooled odds ratio of 1.9 (95%CI: 1.1-3.2) compared to the general population. The pooled estimates of fracture incidence were 11.3 per 1000 person-years (95% CI: 7.9-14.5) with incidence rate ratio (IRR) of 1.5 (95% CI: 1.3-1.8) compared to the general population. Risk factors for fracture incidence were older age (aHR 1.4, 95% CI: 1.3-1.6), smoking (aHR 1.3, 95% CI: 1.1-1.5), HIV/HCV co-infection (aHR 1.6, 95% CI: 1.3-1.9), and osteoporosis (aHR 3.3, 95% CI: 2.2-5.1).
3. Bedimo R, Maalouf NM, Zhang S, Drechsler H, Tebas P. Osteoporotic fracture risk associated with cumulative exposure to tenofovir and other antiretroviral agents. AIDS. 2012;26:825-31.








|
| |
|
 |
 |
|
|