 |
 |
 |
| |
CAB LA FOR HIV PREVENTION IN AFRICAN
CISGENDER FEMALE ADOLESCENTS (HPTN 084-01)
|
| |
| |
CROI 2023 Feb 20-23
Sybil Hosek, PhD
Stroger Hospital of Cook County/CORE Center & RUSH University
Chicago, IL, USA
Hosek S, hamilton e, Ngo J, Jiao Y, Hanscom B, Delany-Moretlwe S, Mgodi N, Siziba B, Naidoo I, Mirembe Gati B, Kamira B, Marzinke M, McCoig C, Spiegel H, Stranix-Chibanda L, for the
HPTN 084-01 Study Team
program abstract
Background: While adolescents represent a key population in need of HIV prevention worldwide, daily oral TDF/FTC pre-exposure prophylaxis (PrEP) confers adherence challenges that limit effectiveness. This single arm, open-label Phase 2b safety study (HPTN 084-01) examined the safety, tolerability and acceptability of long-acting injectable cabotegravir (CAB-LA) among adolescent cisgender females in South Africa, Uganda and Zimbabwe.
Methods: Sexually active, HIV-uninfected females under 18 years of age, willing to use reliable long-acting contraception and weighing at least 35kg were eligible to participate. Parental/guardian consent was required for participation unless youth were considered emancipated minors. Step 1 involved a 5-week oral CAB 30mg QD lead-in, followed by 5 intramuscular gluteal injections of 3 mL (600 mg) of CAB-LA administered at weeks 5, 9, 17, 25 and 33 (Step 2). Participants then switched to daily oral TDF/FTC for 48 weeks of follow-up (Step 3) or opted to continue CAB-LA via open label extension (HPTN 084 OLE). We present safety data, injection tolerability, product acceptability, and adverse events (AEs) of special interest (Table 1).
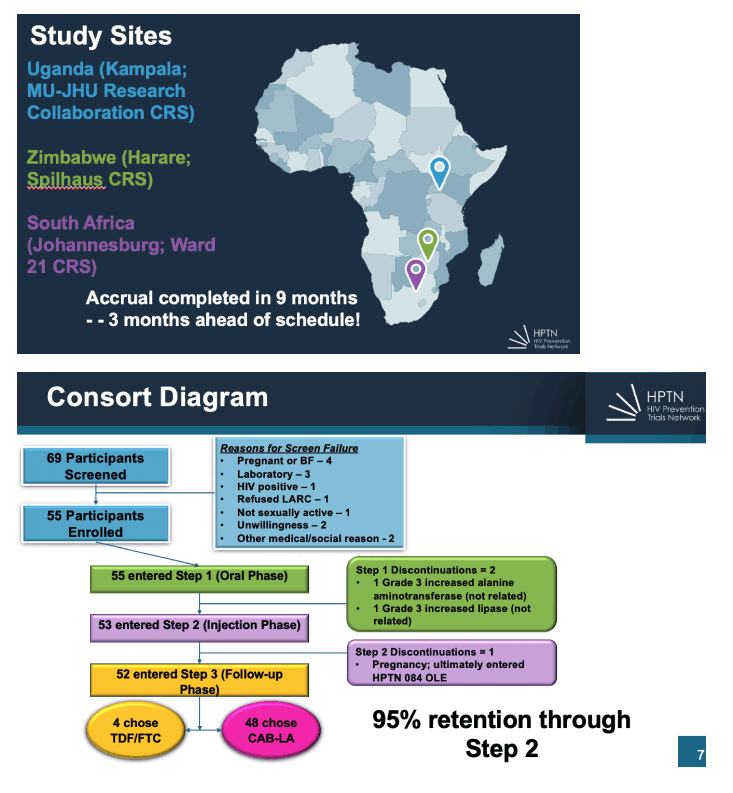
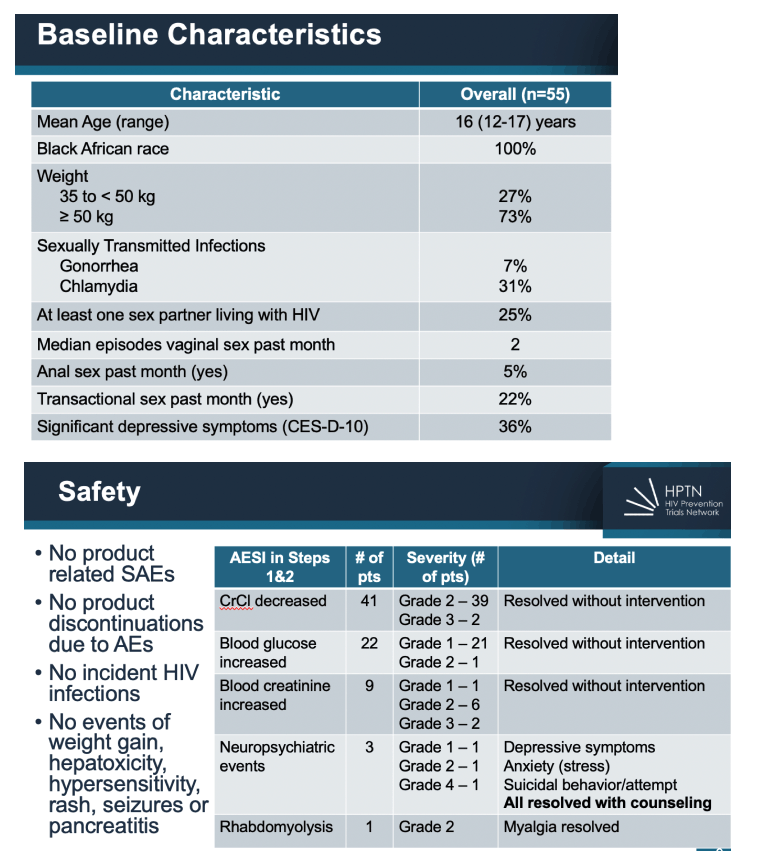
Results: From November 2020-August 2021, 69 individuals were screened and 55 were enrolled (mean age=16, age range 12-17; 100% Black African). At baseline, most had a primary sexual partner in the past month (71%),
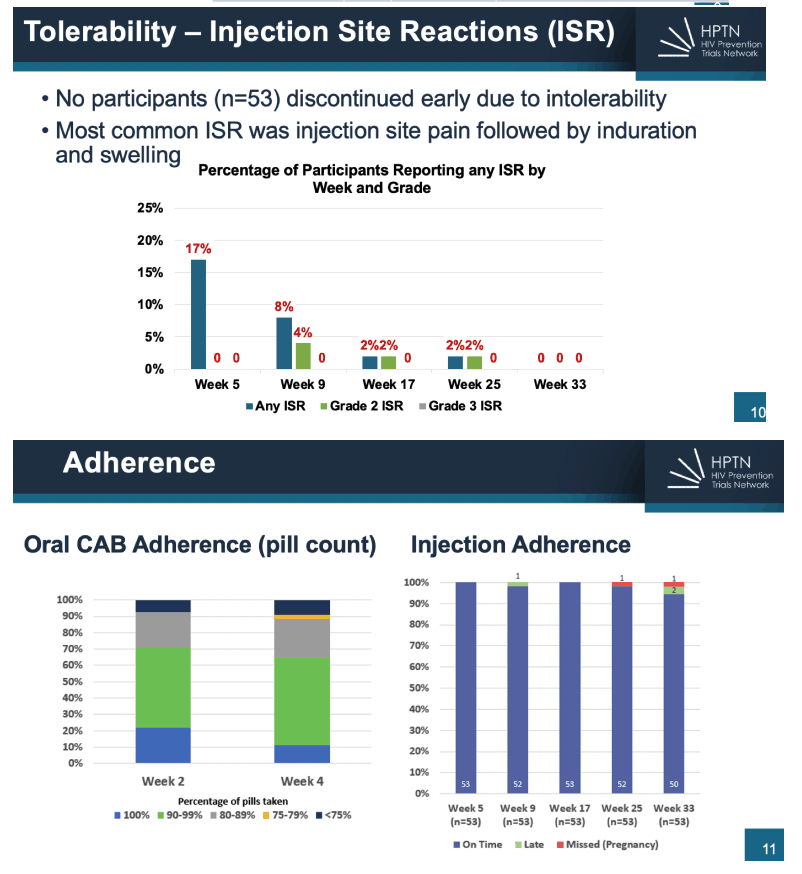
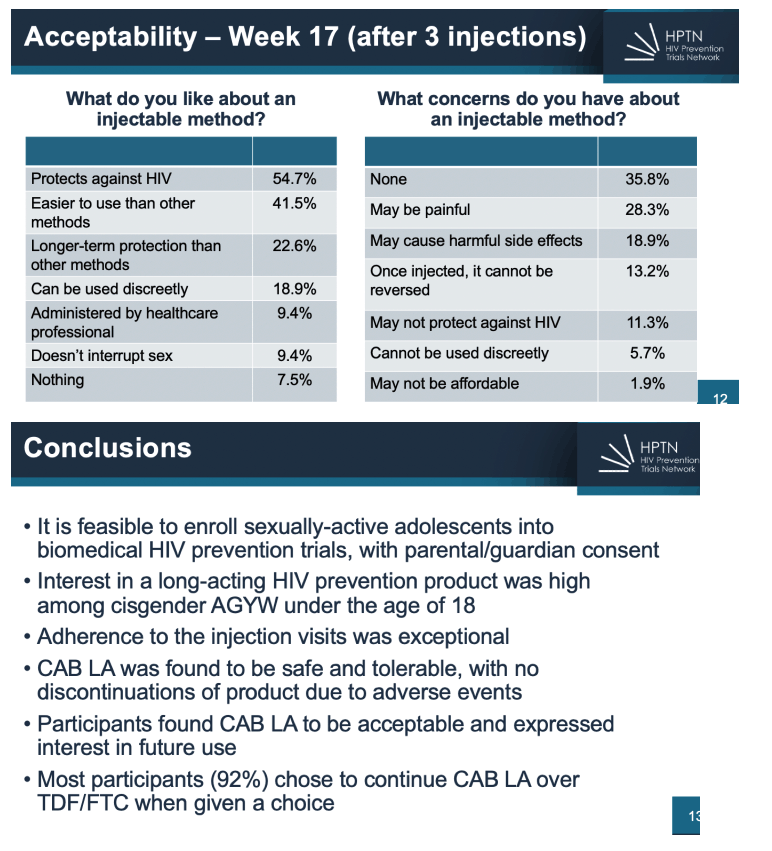
22% reported transactional sex, 31% had genitourinary chlamydia and 7% had gonorrhea. Fifty-two (95%) of participants completed Step 1 and 2; two participants discontinued during Step 1 for AEs unrelated to CAB and 1 participant who started Step 2 stopped CAB-LA after 3 injections due to incident pregnancy. There were no product-related serious AEs nor discontinuations from product due to AEs. No one acquired HIV on study. CAB-LA injections were well tolerated overall. Fourteen participants (26%) experienced 20 injection site reactions (ISR) - all Grade 1 or 2. For acceptability, 62% of participants reported via survey that they would consider using CAB-LA for HIV prevention in the future and 94% of participants went on to continue CAB-LA via HPTN 084 OLE.
Conclusion: CAB LA for HIV PrEP was found to be safe, tolerable and acceptable to sexually active adolescent females under the age of 18 in 3 African countries. Concurrent evaluation of CAB-LA for adolescents and adults has allowed
for simultaneous licensure in several countries to date, expanding the HIV prevention options available to youth in circumstances of heightened risk.



|
| |
|
 |
 |
|
|