 |
 |
 |
| |
Second-Line DTG + DRV/r Controls HIV Better Than DRV/r + 2 NRTIs
|
| |
| |
30th CROI, Conference on Retroviruses and Opportunistic Infections, February 19-22, 2023, Seattle
Mark Mascolini
After a first-line nonnucleoside (NNRTI) failure, dolutegravir (DTG) plus darunavir/ritonavir (DRV/r) or DTG plus XTC/tenofovir disoproxil (TDF) proved virologically noninferior to the standard of care (DRV/r + two nucleos(t)ides [NRTIs]) after 48 weeks in a large international trial, D2EFT [1]. DRV/r + DTG met criteria for virologic superiority to the standard of care at 48 weeks, but weight gain could be a problem with this novel combination. (XTC means either 3TC or FTC).
In April 2017 researchers launched D2EFT, a 2-arm study to evaluate DRV/r + DTG as a potent second-line combination simpler than standard-of-care DRV/r + 2 NRTIs. In response to changing therapeutic needs and availabilities, in May 2018 D2EFT added a third arm, DTG + XTC/TDF, which has an immediate advantage over DRV/r + DTG: current coformulation. To give the trial adequate statistical power when they added this third arm, the investigators enlarged the study population. The bigger trial did not aim to compare DTG + DRV/r with DTG + XTC/TDF.
Study participants had to be older than 18 and to have virologic failure of a first-line combination including an NNRTI plus two NRTIs. They could not have used a protease inhibitor or an integrase inhibitor, could have no significant comorbidity or active coinfection, and could not be pregnant or breast feeding.
The three-arm trial population enrolled 831 people in 14 countries in Central and South America, Africa, and South and Southeast Asia. Study participants averaged 39.4 years in age, 45.7% were men, 68.6% black, and 25.4% Asian. Median starting CiD4 count stood at 206 and median viral load at about 16,000 copies.
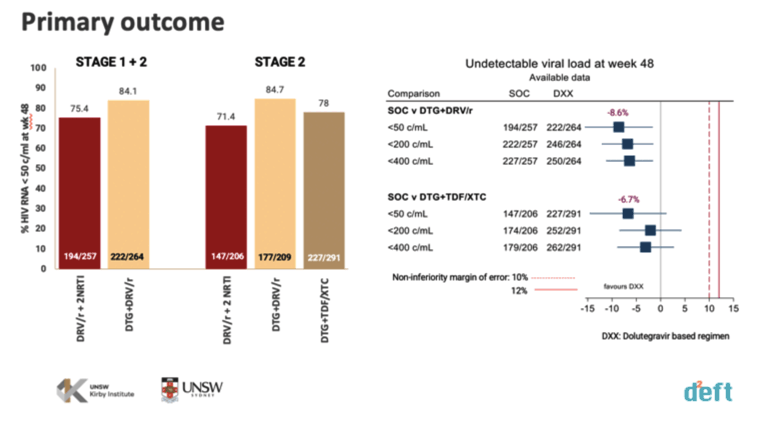
A modified intention-to-treat analysis excluded participants who never started their assigned antiretroviral regimen. After 48 weeks of treatment, 71.4% in the standard-of-care arm (DRV/r + 2 NRTIs) had a viral load below 50 copies, compared with 84.7% in the DTG + DRV/r group and 78% in the DTG + TDF/XTC arm.
DTC + TDF/XTC was virologically noninferior to the standard-of-care regimen (percent difference in viral load below 50 copies -6.7, 95% confidence interval [CI] -14.4 to 1.2, P = 0.09). DTG + DRV/r was virologically superior to the standard of care (percent difference in viral load below 50 copies -8.6, 95% CI -15.5 to -1.7, P = 0.02).
Through 48 weeks CD4 gains averaged 56 cells with DTG + DRV/r (P < 0.001 versus standard of care) and 39.9 cells with DTG + 2 NRTI arm (P = 0.01 vs standard of care). Weight gain also proved significantly greater in the DTG + DRV/r group (2.7 kg, P < 0.001 versus standard of care) and in the DTG + TDF/XTC arm (1.7 kg, P = 0.006 versus standard of care).
Forty-six people switched their second-line regimen, 14 because of adverse events, 11 because of pregnancy, 9 because of treatment failure, 4 because of TB, and the rest for other reasons. Among treatment switches due to toxicity, 5 occurred in the standard-of-care group, 3 in the DRV/r + DTG group, and 6 in the DTG + TDF/XTC group. Among 97 serious adverse events, clinicians judged 14 possibly related to treatment.
In addition to its virologic superiority after NNRTI failure, DTG + DRV/r could have other advantages, the researchers proposed. It includes no NRTIs and requires no drug or drug-class recycling. As a result, among people naive to protease inhibitors and integrase inhibitors, as required in this trial, switching to DTG + DRV/r requires no genotyping. In addition, DTG + DRV/r could be coformulated, which could allow a lower cost. But weight gain could be a problem with this integrase/protease-inhibitor duo.
Reference
1. Matthews G, Borok M, Eriobou N, et al. D2EFT: dolutegravir and darunavir evaluation in adults failing first-line HIV therapy. 30th CROI, Conference on Retroviruses and Opportunistic Infections, February 19-22, 2023, Seattle. Abstract 198.
CROI: D2EFT: DOLUTEGRAVIR AND DARUNAVIR EVALUATION IN ADULTS FAILING FIRST-LINE NNRTI THERAPY - (02/23/23)
|
| |
|
 |
 |
|
|