 |
 |
 |
| |
Twice-Yearly Lenacapavir Demonstrates Sustained
Impact on Health-Related Quality of Life in People With HIV
|
| |
| |
- Patient-Reported Outcomes From the CAPELLA Trial Align With the Demonstrated Safety and Tolerability Profile of Lenacapavir -
- Analyses Show Oral Bridging Maintains Virologic Suppression When Subcutaneous Lenacapavir Doses are Missed -
Press Releases
July 24, 2023
FOSTER CITY, Calif.--(BUSINESS WIRE)-- Gilead Sciences, Inc. (Nasdaq: GILD) today presented new data reinforcing the efficacy, safety, and tolerability profile of lenacapavir, including patient-reported outcomes (PRO) from the Phase 2/3 CAPELLA trial. These latest findings underscore the role of lenacapavir, the first long-acting injectable HIV treatment medication administered twice-yearly, as a person-centric therapy option and its transformative potential impact on the future of coordinated HIV clinical care. The data were presented at the 12th International AIDS Society (IAS) Conference on HIV Science (IAS 2023), taking place July 23-26 in Brisbane, Australia.
Using five validated scoring instruments measuring health-related quality of life components including physical and mental health, CAPELLA participants (n=64/72) reported favorable scores at Week 52 and with relative consistency over the time period. These reflect values that are similar to that of the general U.S. population. Separate analyses from the Phase 2/3 CAPELLA and Phase 2 CALIBRATE trials showed weekly oral lenacapavir, when paired with an OBR, led to high rates of virologic suppression and high, efficacious concentrations of lenacapavir in the blood in study participants when the administration of subcutaneous lenacapavir was interrupted.
"Understanding potential real-world use of novel HIV options is essential to helping ensure they have the greatest potential impact," said Jared Baeten, MD, PhD, Vice President, HIV Clinical Development, Gilead Sciences. "The patient-reported outcome data presented at IAS 2023 provide deep insight into the tolerability of lenacapavir, and the oral bridging data helps demonstrate how twice-yearly lenacapavir can fit into people's lives. As we continue development of lenacapavir-based options with the goal to optimize HIV treatment, people's experience and insights are at the center."
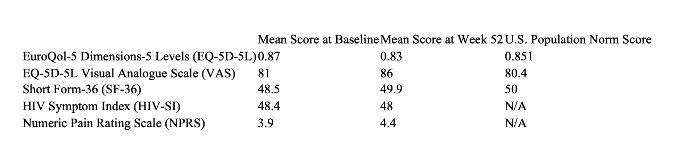
People with HIV who are heavily treatment-experienced may experience symptoms that negatively affect their health-related quality of life, including mobility, self-care, mental health, and physical pain, which could impact their adherence to HIV treatment. Patient-reported outcomes through Week 52 from the CAPELLA study were gathered from five validated instruments that assess quality of life: the EuroQol-5 Dimensions-5 Levels (EQ-5D-5L) index, EQ-5D-5L visual analogue scale (VAS), Short Form-36 (SF-36), HIV Symptom Index (HIV-SI), and Numeric Pain Rating Scale (NPRS). Of the 72 enrolled participants, 64 reported outcomes through Week 52, with notable stability in scores at baseline through Week 52 and generally reflective of the U.S. population norms (see table below). These data demonstrate lenacapavir plus OBR has the potential to be an effective HIV regimen without compromising health-related quality of life.
Additional analyses from CAPELLA and CALIBRATE provided insight on lenacapavir's potential to maintain high rates of efficacious concentrations, virologic suppression, and tolerability when combined with an OBR after missed doses of subcutaneous lenacapavir, and when supplemented with a weekly oral bridging (OB) dose. 79% (57/72) of CAPELLA participants and 78% (82/105) of CALIBRATE participants received an OB dose of lenacapavir (300 mg, once-weekly) on average for 19 weeks, with an adherence rate (by pill count) of ≥95% in most participants.
At Week 10, 20, and Week 30 in a missing equals excluded analysis, 98% (44/45), 97% (30/31), and 100% (10/10), respectively, of participants on OB in CAPELLA who had been virologically suppressed remained virologically suppressed. In CALIBRATE, in a missing equals excluded analysis, 100% of all participants at Week 10, 20, and 30 were virologically suppressed (77/77; 58/58; 5/5, respectively). One CAPELLA participant who missed two non-consecutive oral lenacapavir doses did not maintain virologic suppression during OB. Through the Week 30 OB period, mean plasma concentrations of lenacapavir were maintained above effective concentration (inhibitory quotient-4), indicating 300 mg, once-weekly lenacapavir + OBR may provide concentrations high enough to bridge a missed dose of twice-yearly subcutaneous lenacapavir injection. The use of lenacapavir for oral bridging is not approved by any regulatory authority.
Overall, OB demonstrated a consistent safety and tolerability profile, with treatment-emergent adverse events (AEs) similar to subcutaneous lenacapavir. Two participants in CAPELLA (3.5%) and one participant in CALIBRATE (1.2%) experienced treatment-related diarrhea.
"As clinicians, we seek to understand the real-world constraints people living with the virus may experience with their HIV treatment regimen, and how scenarios when people are unable to receive their next scheduled injection dose could impact their treatment success," said Jean-Michel Molina, MD, Université Paris Cité, Professor of Infectious Diseases and Head of the Infectious Diseases Department at the Saint-Louis and Lariboisière Hospitals. "Results from this analysis provide support for the future potential use of oral therapy with lenacapavir as a treatment strategy to maintain virologic suppression in patients between planned or otherwise missed injection dosing visits in clinical practice."
Twice-yearly lenacapavir is being developed as a foundation for future HIV therapies with the goal of offering both long-acting oral and injectable options with several dosing frequencies, in combination with other antiretroviral agents for treatment or as a single agent for prevention, that could help address individual needs and preferences of people with HIV and people who would benefit from PrEP. The use of twice-yearly lenacapavir for HIV prevention is investigational and the safety and efficacy of lenacapavir for this use have not been established. Twice-yearly lenacapavir is being evaluated as a long-acting option in multiple ongoing and planned early and late-stage clinical studies in Gilead's prevention and treatment research program.
Sunlenca® (lenacapavir), alone or in combination, is not approved by any regulatory authority for any use outside of Australia, Canada, the European Union, Israel, Switzerland, the United Arab Emirates, the United Kingdom or the United States.
Please see below for the U.S. Indication and Important Safety Information for Sunlenca.
There is no cure for HIV or AIDS.



|
| |
|
 |
 |
|
|