 |
 |
 |
| |
Low-Dose Semaglutide Cuts Weight, Triglycerides, and Glucose in HIV MASLD Group
|
| |
| |
CROI 2024 (Conference on Retroviruses and Opportunistic Infections), March 3-6, 2024, Denver
Mark Mascolini
Low-dose once-weekly semaglutide for 24 weeks lowered intrahepatic triglycerides (IHGT) in people with HIV and metabolic dysfunction-associated steatotic liver disease (MASLD) [1]. Lower liver triglycerides correlated with significant improvements in weight, body mass index, waist circumference, fasting plasma glucose, HOMA-IR, HbA1c, and serum triglycerides.
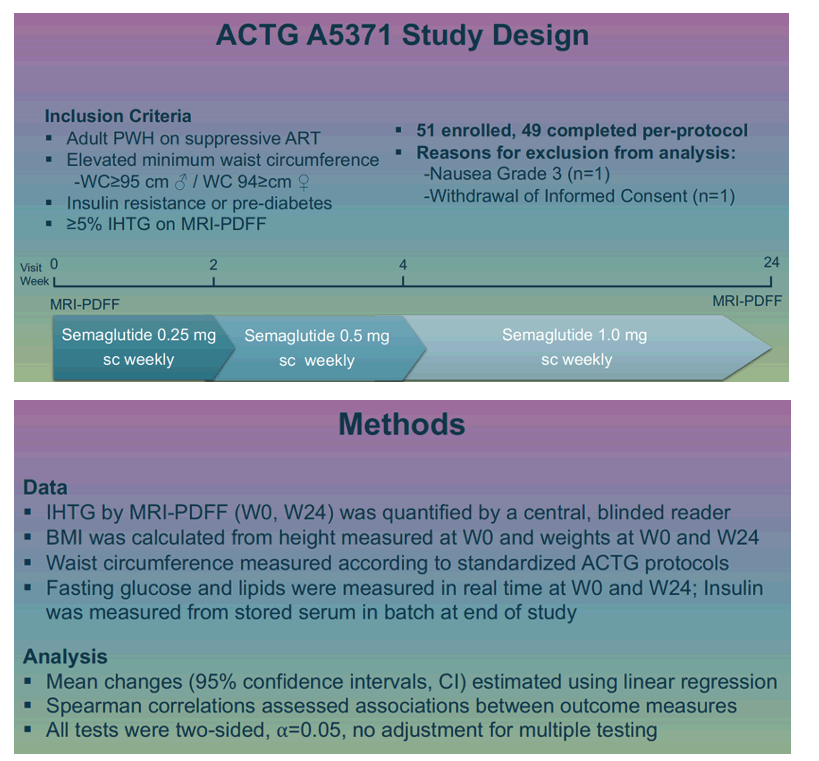
Jordan Lake (University of Texas at Houston) and SLIM LIVER trial collaborators noted that semaglutide leads to cardiometabolic improvements in the general population by lowering weight and quelling systemic inflammation. To see if people with HIV and MASLD derive these same benefits from semaglutide, they conducted a phase 2b, single-arm, open-label pilot study to gauge the impact of semaglutide on intrahepatic triglycerides measured by MRI-PDFF (magnetic resonance imaging-proton density fat fraction) interpreted by a central blinded reader.
To be eligible for the trial (ACTG A5371) adults had to be taking antiretroviral therapy that controlled their HIV infection, have a waist circumference at or above 95 cm in men and 94 cm in women, have insulin resistance or prediabetes, and have 5% or higher intrahepatic triglycerides on MRI-PDFF.
Participants got 0.25 mg of subcutaneous semaglutide weekly for the first 2 weeks, 0.5 mg weekly in weeks 3 and 4, and 1.0 mg weekly from then through week 24. The 49 participants who completed the per-protocol analysis had a median age of 52; 57% were cisgender men, 37% cisgender women, and 6% transgender women. Racial and ethnic proportions were 39% Hispanic, 33% black, 27% white, and 2% American Indian or Alaskan Native. Initial intrahepatic triglycerides averaged 12.7%. Median initial body mass index stood at 35 kg/m2, waist circumference at 114 cm, and CD4 count at 701. Most people (82%) were taking an integrase inhibitor regimen.
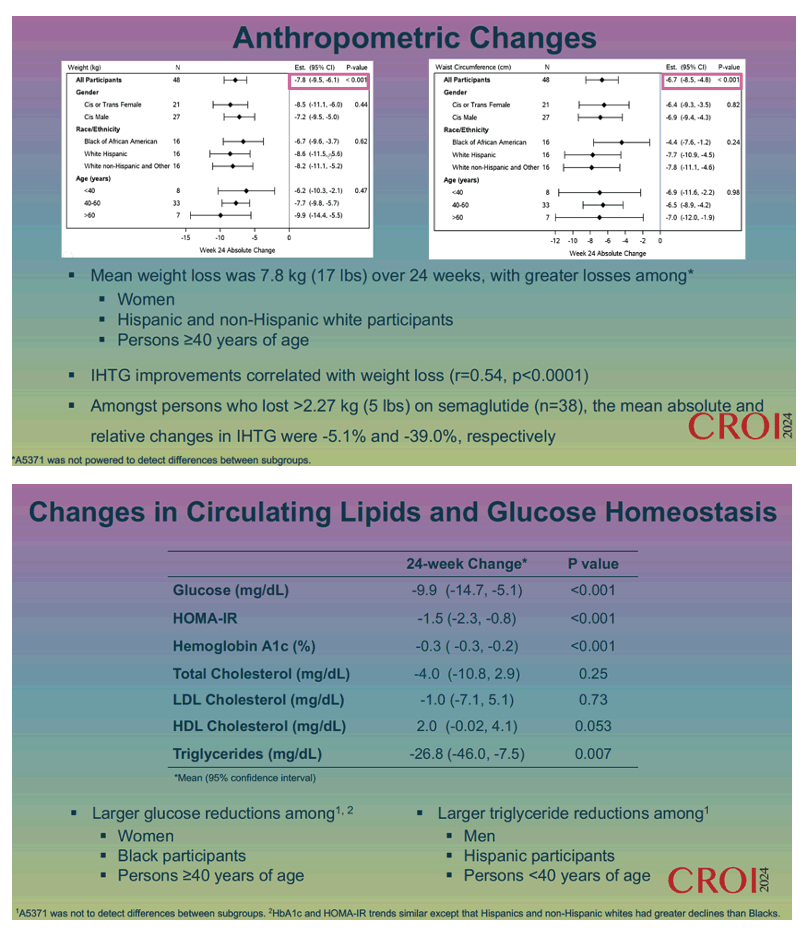
Week-24 absolute change in intrahepatic triglycerides measured -4.2% (95% confidence interval [CI] -5.4 to -3.1, P < 0.001) and week-24 relative change came to -31.3% (95% -39.0 to -23,6, P < 0.001). More than half of participants, 58%, had a 30% or greater relative drop in intrahepatic triglycerides, and 29% had complete resolution of MASLD (absolute intrahepatic triglycerides below 5%). Groups with greater drops of liver triglycerides were women, Hispanic and non-Hispanic whites, and people older than 60 years.
Weight loss averaged 7.8 kg (17 lb) through 24 weeks. Women, Hispanic and non-Hispanic whites, and people 40 or older had the biggest weight drops. Improvements in intrahepatic triglycerides correlated with weight loss (r = 0.54, P < 0.0001). Through 24 weeks of semaglutide therapy, glucose fell 9.9 mg/dL (P < 0.001), HOMA-IR 1.5 (P < 0.001), hemoglobin A1c 0.3% (P < 0.001), and serum triglycerides 26.8 mg/dL (P = 0.007). Glucose declines proved larger in women, blacks, and people 40 or older. Triglyceride drops were bigger in men, Hispanics, and people younger than 40 years.
Researchers recorded only 2 possibly drug-related grade 3 adverse events (nausea and serotonin syndrome) and no grade 4 events.
Lake and colleagues listed unanswered questions about using semaglutide in people with HIV and MASLD as (1) secondary effects (like lean mass loss), (2) durability of treatment benefits, and (3) strategies to maintain benefits.
Reference
1. Lake JE, Kitch DW, Kantor A, et al. Semaglutide reduces metabolic-associated steatotic liver disease in people with HIV: The SLIM LIVER. CROI 2024 (Conference on Retroviruses and Opportunistic Infections), March 3-6, 2024, Denver, Abstract 159.



|
| |
|
 |
 |
|
|