| |
Undertesting & Under Diagnosing of Children with Perinatal HCV
|
| |
| |
Download the PDF here
Frequency of Children Diagnosed with Perinatal Hepatitis C, United States, 2018-2020
A new study was published in January 2024 by the CDC, reported below in depth. This study found:
• Less than one-quarter (427, or 18.8%) of these perinatally exposed infants were tested for HCV infection.
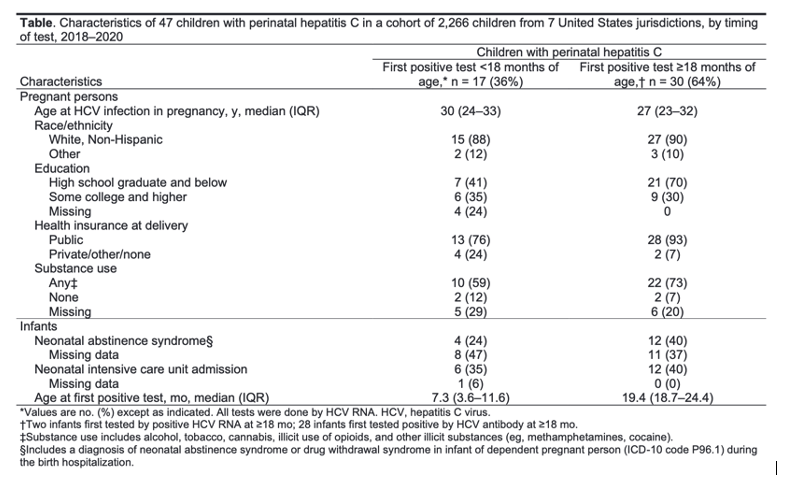
• Less than half of infants expected to have perinatal HCV infection in this cohort were diagnosed - only 36% (47 observed/131 expected)
• 47 of the infants were found to be infected with HCV (2% or 47/2266).
• Using an HCV vertical transmission rate of 5.8%, the researchers estimated that at least 131 of the 2,266 infants should have been infected with HCV-suggesting that an additional 84 children with perinatally acquired HCV had not been diagnosed.
• CDC Recommends universal screening for all pregnant persons during each pregnancy & for all adults >18 at least once
• CDC recommends testing of all perinatally exposed infants for detection of HCV RNA at age 2-6 months
• Studies estimate 5.8% to 7.2% of all perinatally exposed infants & children, and 10.8% risk among poorly controlled HIV coinfected
• Curative HCV DAAs antiviral therapy can be administered at age 3.
overall rates of HCV infections during pregnancy have increased by 20% during 2016-2020 and up to tenfold during 2000-2019
Proper identification of perinatally infected children, referral to care, and curative treatment are critical to achieving the goal of hepatitis C elimination.
Approximately 6%-7% of perinatally exposed children will acquire perinatal HCV infection. Curative DAA therapy is FDA approved for children aged ≥3 years; however, more than one half of perinatally infected children are not tested or linked to care.
In 2020, because of the changing epidemiology of HCV infections in the United States, CDC expanded previous risk-based testing recommendations to include universal screening for all adults aged ≥18 years at least once and for all pregnant persons during each pregnancy (12). Studies have estimated that chronic HCV infection will develop in 5.8%-7.2% of all perinatally exposed (i.e., exposed during pregnancy or delivery) infants and children (13,14), and curative direct-acting antiviral (DAA) therapy can be administered beginning at age 3 years (15,16). A systematic review and meta-analysis of 109 articles found that the risk for perinatally acquired infection from an HCV antibody-reactive pregnant person with detectable HCV RNA was 5.8% (95% CI = 4.2%-7.8%) in the absence of HIV infection and 10.8% (95% CI = 7.6%-15.2%) among those with poorly controlled HIV coinfection (13).
However, most perinatally exposed infants and children are not tested for HCV infection and are not referred for hepatitis C care (17-20); reasons for this might include lack of awareness of perinatal exposure by pediatric providers, lack of regular pediatric care among exposed infants and children, changes in health care providers before the time of HCV testing (recommended at age 18 months), and challenging social circumstances for parents and guardians.
| |
| |
| |
|
|
|