| |
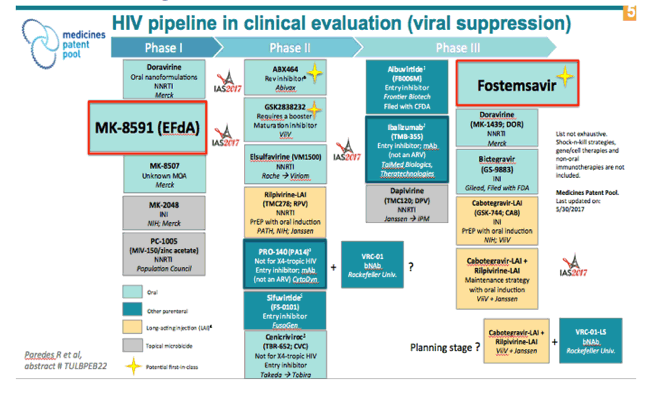
HIV Drug Pipeline
|
| |
| |
IAS: New HIV & HCV Drugs at IAS / Dual ARTs - - (08/09/17)
IAS: Raltegravir 1200 mg Once Daily Versus Raltegravir 400 mg Twice Daily, in Combination With Tenofovir Disoproxil Fumarate/Emtricitabine, in Previously Untreated HIV-1 Infection Through Week 96- - (07/24/17)
IAS: Sub-Study 202094 of SWORD-1 and SWORD-2: Switch From TDF-Containing Regimen to DTG + RPV Improves Bone Mineral Density and Bone Turnover Markers Over 48 Weeks- - (07/26/17)
IAS: A Phase 3 Randomized Controlled Clinical Trial of Bictegravir in a Fixed Dose Combination, B/F/TAF, vs DTG/ABC/3TC in Treatment-Naïve Adults at Week 48- - (07/23/17)
IAS: Phase 3 Randomized, Controlled, Clinical Trial of Bictegravir Coformulated With FTC/TAF in a Fixed-Dose Combination vs Dolutegravir + FTC/TAF in Treatment-Naïve HIV-1-Positive Adults: Week 48 Results- - (07/27/17)
IAS: Gilead Announces Phase 3 Results for Investigational Fixed-Dose Combination of Bictegravir, Emtricitabine and Tenofovir Alafenamide for Treatment of HIV-- (07/23/17)
IAS: Efficacy and safety of switching from boosted-protease inhibitors plus emtricitabine/tenofovir disoproxilfumarate regimens to the single-tablet regimen (STR) of darunavir/cobicistat/emtricitabine/tenofoviralafenamide(D/C/F/TAF) in virologicallysuppressed, HIV-1-infected adults through 24 weeks: EMERALD Study- - (07/25/17)
IAS: Fixed-Dose Combination of Doravirine/Lamivudine/TDF is Non-Inferior to Efavirenz/Emtricitabine/TDF in Treatment-Naïve Adults With HIV-1 Infection: Week 48 Results of the Phase 3 DRIVE-AHEAD Study- - (07/25/17)
IAS: Merck's Investigational NNRTI, Doravirine, Meets Primary Efficacy Endpoint of Non-Inferiority to Efavirenz, Both in Combination with Other Antiretroviral Agents, in Pivotal Phase 3 Trial for Treatment of HIV-1 Infection- - (07/25/17)
IAS: Long-acting intramuscular cabotegravir and rilpivirine in adults with HIV-1 infection (LATTE-2): 96-week results of a randomised, open-label, phase 2b, non-inferiority trial- - (07/24/17)
IAS: Safety, Tolerability, and Pharmacokinetics of Long-Acting Injectable Cabotegravir in Low-Risk HIV-uninfected Women and Men HPTN 077- - (07/26/17)
CROI: Discovery of Novel Potent HIV Capsid Inhibitors with Long-Acting Potential- - (02/16/17)
Monoclonal Antibody Long Acting - LONG-ACTING IBALIZUMAB IN PATIENTS WITH MULTI-DRUG RESISTANT HIV-1: A 24-WEEK STUDY-- (02/16/17)
CROI: Theratechnologies Announces New Data from the Pivotal Phase III Trial of HIV Monoclonal Antibody and Long-Acting Investigational Antiretroviral Ibalizumab- - (02/16/17)
Monoclonal Antibody Suppresses HIV in Half With Multidrug-Resistant Virus- - written by Mark Mascolini - (02/15/17)
FDA Grants Priority Review to HIV Monoclonal Antibody and Long-Acting Investigational Antiretroviral Ibalizumab- - (07/03/17)
CROI: PRO 140 Single-Agent Maintenance Therapy for HIV-1 Infection: A 2-Year Update- - (03/01/17)
Efficacy and safety of long acting HIV fusion inhibitor albuvirtide in antiretroviral-experienced adults with HIV-1: interim 48 week results from the randomized, controlled, phase 3, non-inferiority TALENT study - (11/02/16)
IAS: ABX464 decreases total HIV DNA in PBMC's when administered during 28 days to HIV-infected patients who are virologically suppressed - (07/28/17)
Differential Efficacy of ABX464 and its Primary Metabolite ABX464-NGlc on HIV Replication: Implications for Treatment Strategies to Eliminate Viral Reservoirs - (12/19/16)
HIV Combinectin GSK3732394: A Long-Acting Inhibitor With Multiple Modes of Action- - (10/30/16)
CROI: HIV Attachment Inhibitor Fostemsavir / BMS-663068, GSK3684934- - (02/16/17)
HIV-1 Attachment Inhibitor Prodrug BMS-663068 [GSK3684934 (formerly BMS-663068)] in Antiretroviral-Experienced Subjects: Week 96 Safety Analysis- - (11/02/16)
Pharm Wk: Early Safety, Tolerability and Pharmacokinetic Profile of GSK2838232, a Novel 2nd Generation HIV Maturation Inhibitor, as Assessed in Healthy Subjects- - (06/20/17)
IAS: Resistance Profile of HIV-1 Maturation Inhibitor GSK3532795 / GSK28338232- - (09/07/17)
GSK (formerly BMS) Attachment Inhibitor - IAS: The Effect of Fostemsavir on the Pharmacokinetics of a Combined Oral Contraceptive (OC) Containing Ethinyl Estradiol (EE) and Norethindrone (NE) in Healthy Female Subjects- - (09/07/17)
IAS: Single Doses as Low as 0.5 mg of the Novel NRTTI MK-8591 Suppress HIV for At Least Seven Days- - (07/26/17)
IAS: MK-8591 Protects 8 of 8 Macaques From Rectal SHIV 6 Days After Dosing- - (07/23/17)
IAS: Weekly Oral MK-8591 Protects Male Rhesus Macaques against Repeated Low Dose Intrarectal Challenge with SHIV109CP3- - (07/23/17)
CROI: GS-9131 is a Novel NRTI with Activity Against NRTI-Resistant HIV-1- - (02/23/17)
CROI: Elsulfavirine as Compared to Efavirenz in Combination with TDF/FTC: 48-week Study- (03/02/17)
CROI: A Phase 2 Open-Label Trial of Antibody UB-421 Monotherapy as a Substitute for HAART- - (03/06/17)
New HIV Drugs at CROI / CROI Antiretroviral Treatment Summary / Long Acting HIV Therapies- - (03/31/17)
IAS: New HIV Drugs, Aging, Life Expectancy - (07/26/17)
|
|
| |
| |
|
|
|