| |
"Are we ready for IFN-free treatment regimens?"
|
| |
| |
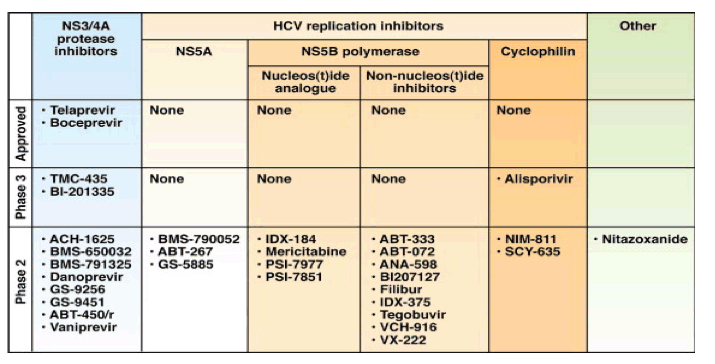
Approved by the FDA in Spring 2011 were the first 2 HCV protease inhibitors telaprevir with 79% SVR rates in clinical study and boceprevir with 70% SVR rate in clinical study for genotype 1 patients, for whom the previous SVR rates in clinical studies were 40% and 25% for African-Americans. The efficacy of oral HCV drugs appears to be the same for African-Americans & caucasians unlike when using peginterferon+ribavirn because the new HCV oral drugs are direct-acting antivirals, not immune-based therapy which is what peginterferon is. In the telaprevir & boceprevir phase 3 studies of African-Americans the SVR rates were 63-66%, lower than overall SVR rate because peginterferon was still in the treatment. But once we have multiple oral HCV drug regimens of 2, 3 or 4 oral HCV drugs the SVR rates are expected to be the same for African-Americans, whites & Latinos. Ongoing now are numerous studies examining interferon-free treatment regimens including some with ribavirin and some without, in both genotype 1 patients & in genotype 2/3 patients. But this is only the beginning. In 2 to 2 1/2 years it's expected 4 new classes of HCV drugs will be FDA approved, making multi-drug combinations possible (2 additional protease inhibitors TMC435, BI201335, BMS790052 (NS5A inhibitor), alisporivir (cyclophillin inhibitor), Gilead's nucleotide PSI-7977). As you can see below there are many studies exploring combinations of new HCV drugs, soon there will be more studies examining more combinations of 3 maybe 4 new oral HCV drugs. The 'proof-of-concept' study has already been done showing 100% SVR with 4 drugs (BMS protease+ BMS NS5A + peg/rbv) but more importantly 4/11 null responders were cured in this study with only these 2 oral drugs & interferon/RBV free therapy. So all these studies below were planned to duplicate this finding, and the results from future studies are likely to be better, these studies are being conducted in treatment-naive patients and the hard-to-treat prior nonresponders called null responders. Researchers will study 12 and 16 weeks therapy for genotype 1 patients to see how much we can shorten therapy, and for some patients with certain regimens I expect 12 or 16 weeks will achieve SVR. Jules Levin
Proof-Of-Concept Study Showing 4/11 null responders cured with interferon/RBV-free therapy with only 2 oral HCV drugs BMS650032 (NS3/4A protease inhibitor)+ BMS790052 (NS5A inhibitor):
Quadruple Therapy With BMS-790052, BMS-650032 and Peg-IFN/RBV for 24 Weeks Results in 100% SVR12 in HCV Genotype 1 Null Responders: original slide presentation at EASL April 2011 Proof of Concept that SVR is Achievable Without Peg/RBV - 4/11 null responder patients achieved SVR w/o Peg/Rbv, with only BMS-790052 (NS5A inhibitor) + BMS-650032 (protease inh) - (02/03/12)
Interferon-Free Treatment Regimens for Hepatitis C: Are We There Yet? Editorial - - (12/12/11)
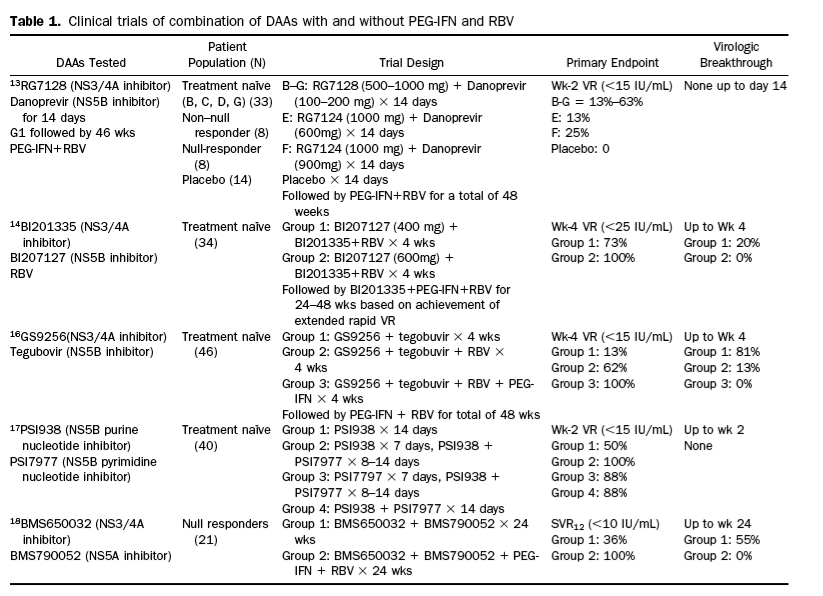
- the INFORM-1 study was the first several years go by Roche to examine interferon-free therapy, involved a combination of an NS5B polymerase inhibitor (RG7128) and an NS3/4A protease inhibitor (danoprevir).
- BI201335 (NS3/4A protease inhibitor)+ BI207127 (NS5B NNRTI polymerase inhibitor) + RBV
- BMS650032 (NS3/4A protease inhibitor)+ BMS790052 (NS5A inhibitor) (+/- RBV)
- TMC435 (protease) + PSI-7977 (+/- RBV)
- BMS790052 (NS5A inhibitor) + PSI-7977 (+/- RBV)
- PSI-7977 (nucleotide) + RBV
- ABT-450 (protease) With Ritonavir (ABT-450/r) When Given Together With ABT-333(NNRTI polymerase) (and Ribavirin (RBV) in Treatment-Naïve and Non-responder Subjects With Genotype 1
- Vertex' telaprevir+VX222 (NNRTI polymerase) +RBV (study recruiting)
- at AASLD Nov 2011, Pharmasset reported 100% SVRs in GT2/3 with RBV+PSI-7977, their nucleotide)
- Alisporivir (cyclophillin inhibitor) + RBV in genotype 2/3
New HCV Drugs at AASLD - (11/21/11)
BI HCV Protease & Other New HCV Drugs - (11/09/11)
Virologic response to an interferon-free regimen of BI 201335 and BI 207127, with and without ribavirin, in treatment-naïve patients with chronic genotype-1 HCV infection: Week 12 interim results of the SOUND-C2 study - (11/08/11)
Efficacy of the Protease Inhibitor BI 201335, Polymerase Inhibitor BI 207127, and Ribavirin in Patients With Chronic HCV Infection - (12/12/11)
Vertex and Alios BioPharma Begin Clinical Studies of Nucleotide Drug Candidates ALS-2200 and ALS-2158 for the Treatment of Hepatitis C - (12/10/11)
Recruiting A Study to Evaluate the Efficacy, Safety, and Pharmacokinetics of ABT-450 With Ritonavir (ABT-450/r) When Given Together With ABT-333 and Ribavirin (RBV) in Treatment-Naïve and Non-responder Subjects With Genotype 1 Chronic Hepatitis C Virus (HCV) Infection
Pharmacokinetics and Pharmacokinetic/Pharmacodynamic Relationship for MK-5172, a Novel Hepatitis C Virus (HCV) NS3/4A Protease Inhibitor, in Genotype 1 and Genotype 3 HCV-Infected Patients - (11/16/11)
Dual Oral Combination Therapy with the NS5A Inhibitor Daclatasvir(DCV; BMS-790052) and the NS3 Protease Inhibitor Asunaprevir(ASV; BMS-650032) Achieved 90% Sustained Virologic Response (SVR12) in Japanese HCV Genotype 1b-Infected Null Responders - (11/08/11)
Activity of danoprevir plus low-dose ritonavir in combination with peginterferon alfa-2a (40KD) plus ribavirin in previous null responders - (04/01/11)
Once-daily alisporivir interferon (IFN)-free regimens achieve high rates of early HCV clearance in previously untreated patients with HCV genotype (G) 2 or 3 - (11/09/11)
TMC435 in Combination with Peginterferon and Ribavirin in Treatment-naïve HCV Genotype 1 Patients: Final Analysis of the PILLAR Phase IIb Study (TMC435-C205) - (11/08/11)
PSI-7977: ELECTRON Interferon is not required for Sustained Virologic Response in Treatment-Naïve Patients with HCV GT2 or GT3 - (11/07/11)
Alisporivir Plus Peg-IFN_-2a/Ribavirin Treatment for Chronic Hepatitis C Genotype 1 Treatment-Naïve Patients Shows Superior Sustained Virologic Response Irrespective of IL28B Genotype and High Barrier to Resistance (12/07/11)
Host targeting antiviral (HTA) alisporivir presents a high barrier to resistance both in vitro and in HCV patients with no cross-resistance to DAAs (12/07/11)
|
|
| |
| |
|
|
|